How white blood cells jump into action in response to foreign microbes
Pro-inflammatory molecules in the blood are essential for fighting off microbial invaders. But too much of these immune-signaling factors, and the body can go into septic shock. A team from the A*STAR Singapore Immunology Network has now elucidated the mechanism by which bacterial pathogens can rapidly trigger the processing of a key pro-inflammatory protein into its active form.
This discovery "offers new drug targets for acute and chronic inflammatory disorders, including sepsis," says Alessandra Mortellaro, the A*STAR immunologist who led the research.
The body's immune system reacts to microbial infection through a group of white blood cells that release immune-signaling molecules known as cytokines, including one called interleukin-1β (IL-1β). Yet, this cytokine occurs first in a biologically inactive form, and must be processed by caspase-1, an enzyme which itself is activated by a multi protein complex called the inflammasome.
In most types of immune cells, revving up the inflammasome requires a priming stimulus followed by a second activation signal. But in monocytes—the key mediators of early responses to infection—inflammasome activation happens in a distinctive one-step pathway following exposure to a component of the bacterial wall, known as a lipopolysaccharide (LPS). How this occurs, however, was poorly understood until Mortellaro and her colleagues found out.
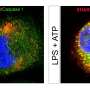
The researchers isolated monocytes from human blood, cultured the cells in their laboratory, and then added LPS derived from the bacterium Escherichia coli. They noticed that two enzymes, caspase-4 and -5, seemed to be expressed in the cells following LPS exposure, and this led to rapid processing of the caspase-5.
Mortellaro and her team then blocked the activity of these enzymes to show not only that caspase-4 and -5 are essential for cytokine release, but that some of the products of caspase-5 processing and other key molecular players are needed for the one-step activation to work, with no additional cues besides LPS.
Notably, all of the experiments were conducted with human monocytes, not mouse cells as most previous studies had done, and as Mortellaro notes: "There are considerable differences between human and mouse immunity."
Since septic shock results from the flood of cytokines like IL-1β that can follow an infection, Mortellaro hopes that blocking the one-step activation pathway she teased apart could have therapeutic value in patients with sepsis. And there could be applications in other disease contexts, too. "It would be interesting to investigate this pathway in a context of metabolic and chronic inflammatory diseases," Mortellaro says.
More information: Elena Viganò et al. Human caspase-4 and caspase-5 regulate the one-step non-canonical inflammasome activation in monocytes, Nature Communications (2015). DOI: 10.1038/ncomms9761