A new mode of communication between donor and host photoreceptors in retinal dystrophy
UK eye researchers with funding from Fight for Sight have discovered a new means of communication between transplanted donor photoreceptors developed from stem cells and the degenerating photoreceptors of the host retina. The results, published in Nature Communications, show that this previously unreported mechanism of interaction between host-donor photoreceptors, termed 'material transfer', plays a major role in the rescue of visual function.
In progressive retinal conditions such as retinitis pigmentosa and macular dystrophy, the light-sensitive photoreceptor cells degenerate over time. As the photoreceptors lose their ability to respond to light, vision becomes increasingly poor.
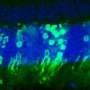
Previous research in adult mice has shown that cell transplantation is a promising potential treatment. Following transplantation of donor photoreceptors tagged with a fluorescent genetic marker, cells containing the same fluorescent marker have been observed in the correct position in the retina. These tagged cells contain all of the proteins they need to function and tests of cell and retinal function and of mouse behaviour suggest that these cells behave as healthy photoreceptors would be expected to and can rescue some aspects of vision.
Until now it was thought that this rescue happens when donor photoreceptors migrate to the receiving retina and integrate into it, replacing the degenerated cells. While these new results confirm that this does happen, they show that the rescue is actually largely due to this newly identified process of material transfer.
Dr Rachael Pearson of the UCL Institute of Ophthalmology, who led the study says:
"Although we do not yet know the cellular mechanism underlying material transfer, we think that 'packages' of genetic and cellular molecules such as RNA and/or proteins are released from donor cells and are then taken up by the degenerating photoreceptors. This process seems to give the ailing cells enough of the proteins they need to recover and get back to work, at least for a period of time, either directly or by providing the genetic instructions to produce them.
"Past reports of material transferred between other types of cell (usually stem cells) have involved donor and recipient cells permanently fusing together, allowing them to share their contents, including their DNA. Photoreceptors, however, show none of the typical signs of donor-recipient cell fusion."
Professor Robin Ali of the National Institute for Health Research (NIHR) Biomedical Research Centre (BRC) at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology, co-senior author on the paper, says:
"From this experiment, we conclude that we have identified an exciting, new and previously unreported physical method of communication between donor and recipient photoreceptors. So far, material transfer has only been shown in mice, and further work will need to be carried out to determine if the same mechanism occurs between the photoreceptors of other species, including humans.
"However, these findings do highlight that cell transplantation into the retina is much more complex than previously thought and provides an important new understanding that will help inform the development of techniques to optimise integration of donor photoreceptors."
Dr Dolores M Conroy is Director of Research at Fight for Sight. She said:
"Material transfer between donor and host photoreceptors is certainly an exciting discovery. Some degenerative retinal conditions are currently untreatable as the underlying cause is not yet known. For these conditions in particular, and if we can determine the mechanism, material transfer may offer a way to offer the degenerating retina a means of repair that can rescue vision to some degree.
"In the meantime, transplanting donor cells that can connect to the host retina remains a promising approach to treating advanced and end-stage retinal dystrophy, in which too few photoreceptors remain for material transfer to occur."
More information: Pearson, R. A. et al. Donor and host photoreceptors engage in material transfer following transplantation of post-mitotic photoreceptor precursors. Nature Communications 7, 13029 (2016). dx.doi.org/10.1038/NCOMMS13029