Startup develops late-stage prostate cancer therapy that could increase patient survival rates

A biomedical startup that licensed a Purdue University technology has developed a late-stage prostate cancer therapy that could provide an alternative to current hormone therapies that are known to develop resistance after prolonged use.
Ji-Xin Cheng, a professor in Purdue's Weldon School of Biomedical Engineering; Junjie Li, a postdoc research fellow in the Weldon School of Biomedical Engineering; and Timothy L. Ratliff, a professor in Purdue's College of Veterinary Medicine and director of the Purdue University Center for Cancer Research, co-founded Resarci Therapeutics LLC to further develop the technology.
Cheng said that resistance to current hormone therapies is one of the biggest challenges patients face in treatment.
"Hormone therapies have a goal to reduce male hormones, called androgens, in the body, or to stop them from affecting prostate cancer cells," he said. "Almost 100 percent of cancer patients will eventually develop a resistance to hormone therapies. Every year in the United States around 32,000 new cancer cases become resistant, lessening the likelihood of survival."
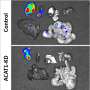
Resarci Therapeutics has developed a new therapeutic strategy by targeting the cholesterol metabolism instead of the androgen pathway.
"By targeting the cholesterol metabolism, which is specific to cancer cells and independent of the hormone signaling pathway, we are able to eliminate the hormone resistance," Li said. "We target the aberrant cholesterol metabolism using an inhibitor of cholesterol esterification enzyme ACAT-1, named avasimibe. Avasimibe selectively kills cancer cells by preventing the cholesteryl ester accumulation and inducing free cholesterol related toxicity in cancer cells."
Ratliff said the company has demonstrated the ability of the inhibitor avasimibe to overcome the resistance to hormone therapy in mouse models and is looking to further improve their product in future trials.
"We want to improve the formulation of our product, test it in preclinical settings and launch an early-stage clinical trial," he said. "We are applying for funding from NIH and other agencies but are also looking for investors or potential partnerships with pharmaceutical companies."