Generating brown fat cells from bone marrow-derived stem cells could transform the treatment of metabolic disorders
A renewable source of brown fat cells, and a simple method for generating them in cell cultures, has been discovered by A*STAR researchers and could help with the development of personalized therapies for metabolic diseases.
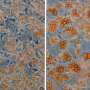
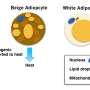
There are two main types of fat cells, or adipocytes, in the body; white adipocytes that store energy, and brown adipocytes that burn energy and generate heat. Babies have an abundance of brown adipocytes as they help infants to keep warm, but recently scientists have discovered the cells also exist in small quantities in adults. By encouraging the body to use, rather than store, excess energy, brown adipocytes could prove useful in the engineered modulation of energy consumption and the regulation of blood sugar levels – two mechanisms that can fail in metabolic disorders such as obesity and diabetes.
Until now, there has been no safe, reliable method of generating large quantities of brown adipocytes to explore their therapeutic potential. A new technique, developed by Michael Raghunath, Cedric Badowski and co-workers at the A*STAR Institute of Medical Biology together with scientists at the National University of Singapore (NUS), hinges on unlocking the potential of mesenchymal stem cells taken from human bone marrow (bmMSCs) to differentiate into brown adipocytes.
"We wanted to determine which fat cells were present in adult bone marrow, because this hadn't been fully investigated before," explains Michelle Lee, a researcher on the project. "To our surprise, we found brown adipocytes present, and discovered that the bone marrow environment could trigger bmMSC differentiation into brown fat cells. We decided to emulate that environment in the laboratory."
When stem cells are taken out of the body and placed in cultures, they lose the crowded, intricate microenvironment that enables them to generate a protective extracellular matrix and function correctly. The team used a technique called 'macromolecular crowding' to mimic the microenvironment and encourage bmMSC differentiation. By adding extra molecules to bulk out the culture, Lee explains, there is an increase in reaction mechanisms within the culture that allows the cells to secrete and remodel the extracellular matrix.
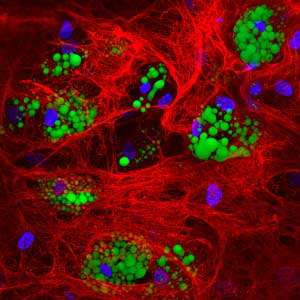
Using cutting-edge microscopes to visualize the processes, the researchers found that macromolecular crowding created a cocoon of collagen around the stem cells, greatly enhancing matrix signaling and stimulating large quantities of viable, fully-functional brown adipocytes from bmMSCs (see image).
"Once the potential for this technology is fully explored, we could transform the treatment of metabolic disorders and provide considerable benefits for society," says Badowski.
More information: Michelle H. Lee et al. ECM microenvironment unlocks brown adipogenic potential of adult human bone marrow-derived MSCs, Scientific Reports (2016). DOI: 10.1038/srep21173