Microbubbles and ultrasound open the blood-brain barrier to administer drugs

The impassable blood–brain barrier prevents microorganisms from entering the brain; however, it also blocks medicines that could help treat Parkinson's, Alzheimer's and other neurodegenerative diseases. Now, a Spanish physicist and other researchers at the University of Columbia (USA) have succeeded in embedding these substances in tiny lipid bubbles in such a way that ultrasound can be used to release them into the specific area of the brain where they are needed.
A defence mechanism as sophisticated as the blood–brain barrier, which protects our brains from viruses, bacteria and fungi that filter into our blood, can become the body's worst enemy when it comes to treating certain illnesses. This "wall" is an impassable obstacle for 98 percent of drugs, which it treats as pathogens and blocks from passing from patients' bloodstream into their brain.
Scientists have for years been trying to break through this natural barrier, but almost none of the techniques developed to date (such as intracranial injections, gene therapy and the chemical modification of the medicine) have been able to transport the drug without affecting the rest of the body and with reversible action, in which the barrier opens and closes very rapidly.
The only method that has been capable of overcoming all these conditions is based on the use of ultrasound after drugs are administered with an independent intravenous injection of lipid-coated gas microbubbles, which are completely innocuous.
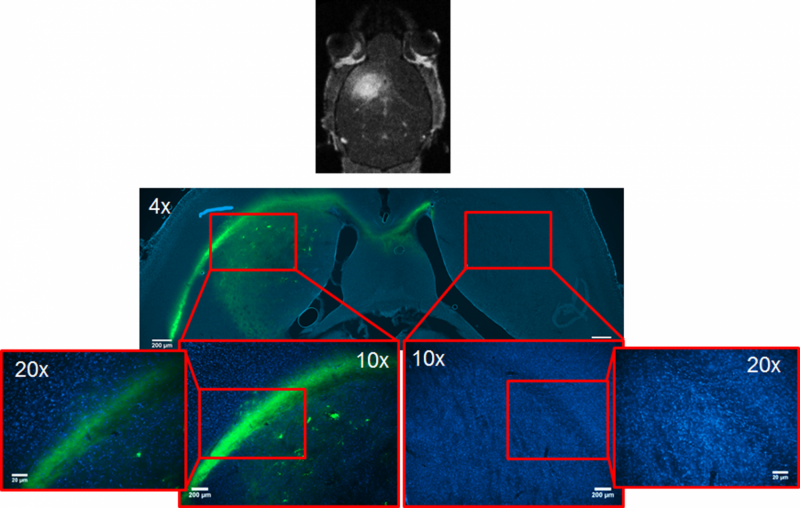
Like a laser, the ultrasound is focused on a specific region of the brain, where the microbubbles begin to oscillate and increase in size due to their interaction with the acoustic waves. When these tiny bubbles reach the critical size of 8 microns, the blood–brain barrier opens, allowing the medicine circulating in the blood to pass through.
This technique has been used successfully for over 10 years, but it does have a disadvantage: The drugs move through the entire circulatory system and reach organs where they are not needed, causing adverse effects. However, it appears that scientists at the Ultrasound Elasticity Imaging Laboratory (UEIL) at New York's Columbia University have found a solution.
"We've made a step forward by incorporating the substance into the lipid coating of the microbubbles. This makes the substance adhere to the microbubbles and prevents it circulating freely through the body," said physicist Carlos Sierra, a UEIL researcher.
With this technique, the microbubbles flow through the entire body with the potential drug adhered to them. The drug is only released in the area of the brain where it is required, where the effect of the ultrasound causes them to rupture and open the blood–brain barrier. "It does all this while being non-invasive, reversible and completely safe," Sierra said.
From experiments on mice to human trials
So far, the researchers have proven the efficacy of their technique on mice. They chose a fluorescent molecule called 5-dodecanoylaminofluorescein and confirmed that it was reaching the brain without affecting other parts of the animal. At the same time, they identified the acoustic pressure thresholds from which the substance is guaranteed to reach its target in vivo.
"Defining these parameters means we can think about how to transfer the technique to human patients, although it has to be tested on monkeys first," the researcher explained, adding that "it could be applied to diseases like Parkinson's, Alzheimer's, Huntington's disease, brain tumours, strokes, multiple sclerosis and amyotrophic lateral sclerosis, where we expect to see a very significant rise in the efficacy of treatment and a considerable reduction in side-effects."
The team has now started administering potentially therapeutic compounds for treating Parkinson's disease "with promising initial findings," Sierra said, concluding that "the success of this technique in mice, and even in monkeys, can't guarantee it will be effective in people, but if we continue to get satisfactory results then pre-clinical trials on humans would begin."
More information: C. Sierra et al. Lipid microbubbles as a vehicle for targeted drug delivery using focused ultrasound-induced blood-brain barrier opening, Journal of Cerebral Blood Flow & Metabolism (2016). DOI: 10.1177/0271678X16652630