Retinitis pigmentosa gene discovery may reveal a new route to inherited blindness
Researchers at UCL Institute of Ophthalmology and Moorfields Eye Hospital with funding from Fight for Sight, in collaboration with a team from Baylor College of Medicine in the USA, have discovered a new retinitis pigmentosa gene. The team used gene editing to confirm that faults in receptor expression enhancer protein 6 (REEP6) cause 'rod' photoreceptor cell degeneration. Their results point to a biochemical pathway that was previously unknown to be important for the retina.
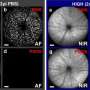
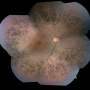
Retinitis pigmentosa (RP) is the most common form of inherited retinal dystrophy, affecting 1 in 4000 people. In RP, light-detecting photoreceptor cells in the retina at the back of the eye degenerate and die over time. It starts with the photoreceptors that are active in low light – the rods – and may later progress to the 'cones' used for seeing colour and detail.
Exactly how RP develops will depend on the specific gene or fault involved. To date there are at least 58 genes linked to autosomal recessively inherited RP, but an estimated 1 in 4 people with RP do not yet have a precise diagnosis. Autosomal recessive conditions are those caused by inheriting two faulty copies of a gene, one from each parent.
In the current study, the research team used whole genome sequencing to identify 6 different faults in REEP6 in people with RP from five unrelated families in the UK and USA. The faults were identified in both copies of the gene in keeping with an autosomal recessive inheritance pattern.
Professor Mike Cheetham at UCL Institute of Ophthalmology was the study's UK lead. He said:
"REEP6 encodes a protein that has not been studied intensively before. We were able to show in human photoreceptors derived from patient stem cells that REEP6 is highly active in rod photoreceptors. We were also able to confirm using CRISPR-Cas9 gene editing that introducing one of the REEP6 variations associated with RP into mice led to symptoms that closely mimic the pattern of photoreceptor degeneration we see in these patients.
"Although we know little about the precise function of REEP6 inside rods our results are interesting and important because the related genes we do know about have not previously been linked to retinal dystrophy. We now have an accurate genetic model of this form of RP in which to investigate further and have shown the usefulness of gene editing to study genetic diseases."
Dr Dolores M Conroy is Director of Research at Fight or Sight. She said:
"We are very pleased to see that more families with inherited retinal blindness are able to have a specific genetic diagnosis. This is both the start of better information around which to plan the future and an important first step on the road to developing treatment. The collaborative working between major centres of genetic research as supported by the RP Genome Project co-funded by Fight for Sight and RP Fighting Blindness is progressing as we intended."
More information: Arno et al., Mutations in REEP6 Cause Autosomal-Recessive Retinitis Pigmentosa, The American Journal of Human Genetics (2016). dx.doi.org/10.1016/j.ajhg.2016.10.008