Researchers strengthen the case for sexual transmission of Zika virus
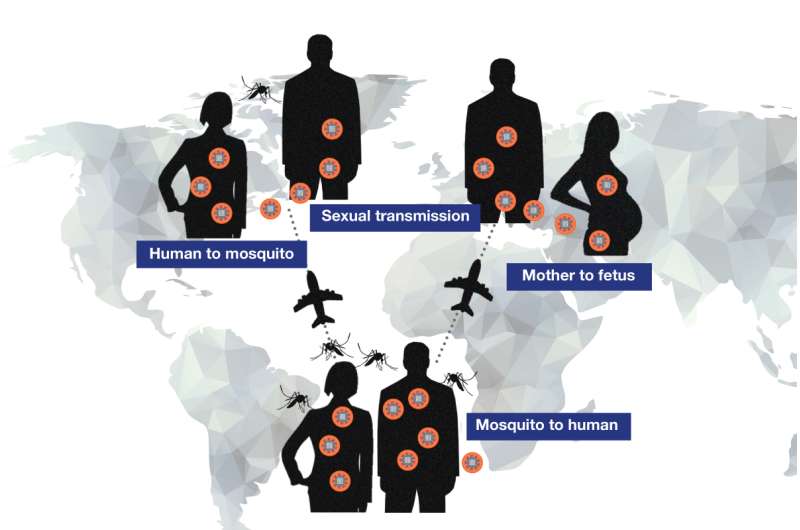
Aedes aegypti mosquitoes harboring parasitic Zika virus (ZIKV) are the primary transmitters of virus to humans, potentially causing catastrophic congenital microcephaly in babies born to women bitten by infected mosquitoes. But confirmation earlier this year by the Centers for Disease Control and Prevention (CDC) that ZIKV can also be sexually transmitted raised new alarm that virus could be passed between sexual partners in venues far from mosquito habitats.
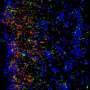
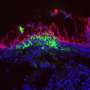
Now La Jolla Institute for Allergy & Immunology (LJI) investigator Sujan Shresta, Ph.D., employs two different mouse models to confirm that live ZIKV placed directly in the vagina infects the mouse's reproductive tract, replicates, moves into the bloodstream, and causes clinical signs of disease. Intriguingly, that study published in the December 20, 2016 issue of Cell Reports, also reports that the stage of the reproductive cycle during which a female mouse is exposed to virus determines vulnerability to infection. If applicable to humans, this discovery has public health implications for virus transmission to a population of great concern, women of child-bearing age.
"Currently, almost all of our efforts in terms of Zika prevention focus on mosquito control," says Shresta, an associate professor in LJI's Center for Infectious Disease. "Our new work begs clinicians to also address whether sexual transmission of the virus constitutes a small or large proportion of cases."
Investigators knew that virus hides in semen of men who contract ZIKV from mosquitoes and that virus is transmitted vaginally in rodent models. But the biological questions—what cells are infected, how stable the virus is in bodily fluids—were unanswered. Shresta's group began to explore them by placing live ZIKV in the vaginas of female mice that had been genetically engineered to be immunocompromised.
But before the procedure, they treated mice with hormones to create two groups that differed with regard to where they were in their menstrual cycle. Dramatic differences emerged post-infection: mice infected in the diestrus or in between phase became progressively sick, lost weight, and died in 2-3 weeks, as one might predict in these mice. Remarkably, the same strain of immunocompromised AG129 mice infected in estrus phase showed no sign of disease.
William Weihao Tang, the study's first author, calls this one the paper's most intriguing findings. "The strain of mice we used, called AG129, were originally engineered to be extremely vulnerable to infection," he says. "But even these mice, when infected in estrus phase, appeared completely resistant to virus. That surprised us."
Shresta says that a caveat is that responses in mouse strains like AG129, which were purposely engineered to serve as a "lethal" model of infection, must be tested in mice with greater immune function. "For science to be relevant to humans, we always confirm results in the most 'immunocompetent' mouse that better reflects a normal human immune system."
To do that, her team repeated experiments in an entirely different type of engineered mouse, one only moderately susceptible to infection, which scientists call a "non-lethal" model. When infected in diestrus phase, those mice lost weight and exhibited clinical signs of disease but, unlike their AG129 counterparts, eventually recovered. However, just like the AG129 mice, when infected in estrus phase "non-lethal" mice showed no sign of Zika-like disease.
This trend was reflected in other experimental outcomes. For example, in both lethal and non-lethal strains, viral RNA, which serves as direct evidence of virus, persisted in the vaginal canal sometimes as long as 10 days post-infection in diestrus. By contrast, viral RNA disappeared three days after infection in estrus phase.
Virus persistence in vaginal fluids may account for why diestrus-infected mice become sick, regardless of mouse strain, yet the molecular or cellular basis for susceptibility remains unclear. Mice analyzed in the study were experimentally synchronized or "staged" at one of two reproductive phases by hormonal injection, which may provide a clue. "Hormones changed the mouse female reproductive tract in ways that either enhanced or protected against sexual transmission," says Tang, although he and Shresta caution it is much too early to generalize mouse findings to humans.
But if similar mechanisms prove relevant to human transmission, they are cause for concern, largely because most Zika-infected men or women show few or no symptoms. Thus they could unwittingly engage in sexual activity resulting in adult disease or even in utero transfer of virus to an unborn child.
Recent CDC "case counts" suggest that thus far that few Zika cases in the US were likely transmitted sexually. But these numbers are estimates, and sexual transmission of ZIKV is taken extremely seriously in other regions, such as South America. In fact, one mathematical modeling study of Baranquilla, Colombia, estimated that as many as 47% of Zika cases reported there emerged from sexual contact.
"In humans sexual transmission may be a bigger deal than has been thought," says Shresta, emphasizing that currently we know very little about this mode of Zika transmission. "We know that in males virus can remain in semen for possibly months, while a man shows no symptoms. During that time he could unknowingly pass it to a sexual partner."
The next step for the Shresta lab is to take advantage of these two mouse models to define immune signals that make mice susceptible to or protected from ZIKV infection. "We are ultimately interested in drugs or vaccines to prevent the disease," says Shresta, who has also used immunodeficient mice as models to study dengue virus infection. "Being able to test interventions in two animal models, one that succumbs to infection and another that recovers, is a plus. Developing vaccines requires access to models representing all scenarios."
More information: "A Mouse Model of Zika Virus Sexual Transmission and Vaginal Viral Replication" Cell Reports, DOI: 10.1016/j.celrep.2016.11.070 , http://www.cell.com/cell-reports/fulltext/S2211-1247(16)31644-8