New device could save glaucoma patients' eyesight
People with high-risk glaucoma will be able to monitor their disease, anywhere, anytime, thanks to a new device developed by an FIU professor.
S.S. Iyengar (Ram Iyengar), director and Ryder professor of the School of Computing and Information Sciences within the College of Engineering and Computing, was just awarded a patent, along with co-inventors, for a tool that provides early intervention for glaucoma. The unnamed device works by directly monitoring intraocular pressure.
Glaucoma is an eye disease in which increased intraocular pressure causes damage to the optic nerve and leads to blindness if left untreated. Diabetics and people above age 50 who suffer from hypertension are at high-risk for developing the disease. According to Iyengar's research, it's estimated that by 2020, 11 million individuals will be blind as a result of glaucoma. Currently, more than 2.2 million Americans have the disease, but only half of them know it. Glaucoma accounts for nine to 12 percent of all cases of blindness in the United States.
For Americans, managing the disease consists of regular monitoring with an ophthalmologist, but for those living in other parts of the world, seeking treatment can be a challenge. If a patient lives in a remote village, for example, it may take hours to reach a doctor, if one is even available, and transportation may not be accessible either.
"In rural communities where there are no doctors, people are going blind," explained Jerry Miller, adjunct faculty and Discovery Lab research coordinator. "If there is no alternative to traveling for a day to get help, most people won't do it."
Iyengar hopes to change that with his device, which is inexpensive and easy to manufacture. "I wanted something low-cost so everyone can afford it," he said. "It's a great device for developing countries. It can cost patients as little as $15-20 once mass produced."
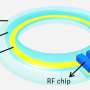
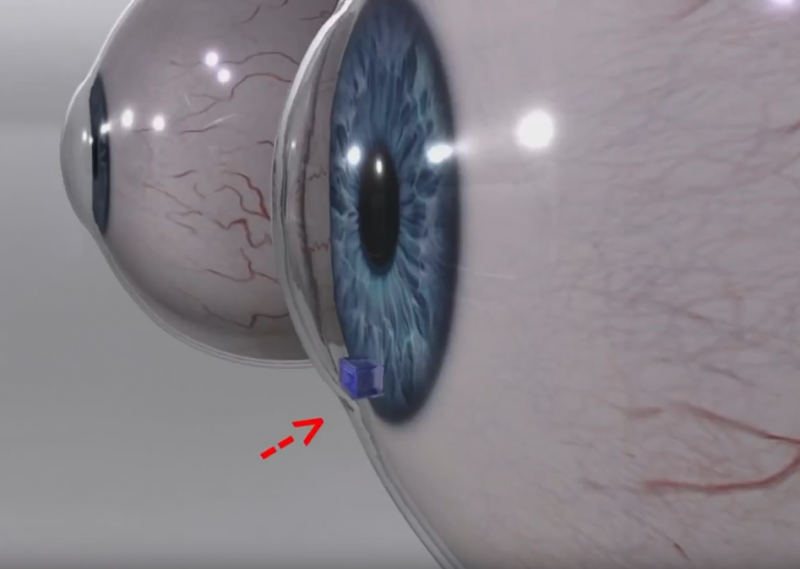
The device, which is the size of a small electronic chip, is surgically implanted in the eye between the iris and the cornea. Patients can keep track of their intraocular pressure, as often as once a week or more if necessary, by simply looking in the mirror. If the device changes color, there is a change in eye pressure, and the person should seek treatment right away to avoid retinal damage.
The device needs no batteries or power supply. Current devices, said Iyengar, require electric power and can be cost-prohibitive. Another competitive advantage is this device eliminates the need to see an ophthalmologist just to take a pressure reading. Patients can monitor their own intraocular pressure at home and seek help only when it registers as high.
Currently, Iyengar is evaluating clinical partners to conduct clinical studies. Eventually, plans are to offer the device as contact lenses.