How two telomere proteins interact with each other and the functional effects of cancer-associated mutations
Scientists at The Wistar Institute have unveiled part of the protein complex that protects telomeres—the ends of our chromosomes. The study, published online in Nature Communications, explains how a group of genetic mutations associated with this protein complex contributes to various cancers.
Telomeres are the protective structures at the end of chromosomes and are essential for the faithful replication and protection of our genome. Defects in telomere function can lead to genomic instability in cancer, while the gradual shortening of telomeres is associated with the aging of human cells. A key component of the telomere protecting mechanism is a multi-protein complex called shelterin. Shelterin protects chromosome ends from triggering DNA damage response mechanisms; it also regulates telomere maintenance and replication by the enzyme telomerase.
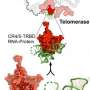
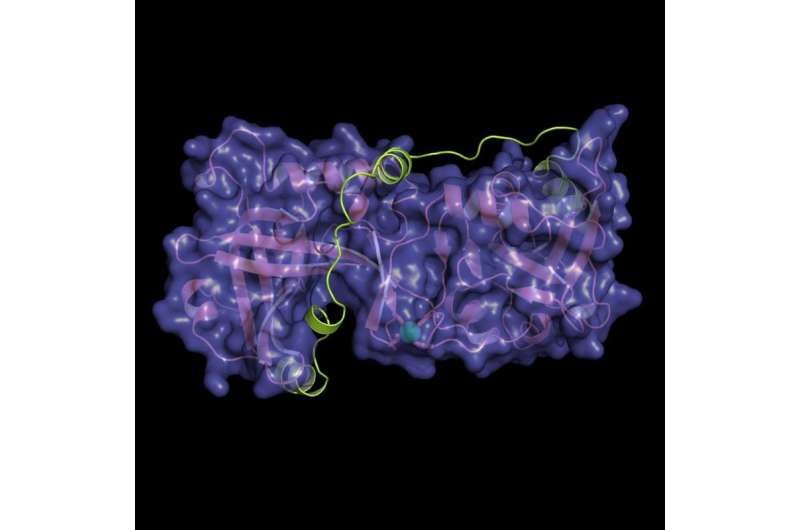
This new study describes the atomic structure of the protein-protein interaction between two subunits of the shelterin complex, POT1 and TPP1. Several gene mutations have been described as affecting portions of POT1 in familial melanoma, glioma and chronic lymphocytic leukemia. Based on a host of structural, biochemical and cell-based data, the study explains how these mutations may contribute to malignant transformation.
"Our research adds a piece of the puzzle toward solving the intricate structure of the shelterin complex, a fundamental player in the maintenance of telomere DNA integrity," said Emmanuel Skordalakes, Ph.D., Associate Professor in the Gene Expression and Regulation Program at Wistar and lead author of the paper. "Based on our data, we suggest that the cancer-associated mutations reported in POT1 affect the integrity of telomeres, leading to chromosomal abnormalities and in turn genomic instability, which is a hallmark of cancer progression. We think that this mechanism works in combination with other fundamental genetic defects characteristic of these malignancies."
The Skordalakes lab used x-ray crystallography to reveal the structure of the POT1-TPP1 complex. The structure was used as a platform to design biochemical and cell-based assays to study the effect of several mutations on this structure. They found that some of these mutations reduce the affinity between the two proteins and, consequently, their ability to bind to telomeric DNA efficiently, whereas others perturb the POT1 structure. They also used imaging techniques to look at different aspects of chromosomes and telomeres and found that these mutations result in defects in telomere length and structure, such as fragile telomeres that tend to break during replication stress, missing telomeres and chromosome fusion between telomeres from two different chromosomes.
This study sheds light on the mechanism by which these cancer-related genetic mutations affect telomere stability, and further research will be necessary to better understand their precise function and how it relates to cancer.
More information: Cory Rice et al. Structural and functional analysis of the human POT1-TPP1 telomeric complex, Nature Communications (2017). DOI: 10.1038/ncomms14928