Cell mechanism discovery could lead to 'fundamental' change in leukaemia treatment
Researchers have identified a new cell mechanism that could lead to a fundamental change in the diagnosis and treatment of leukaemia.
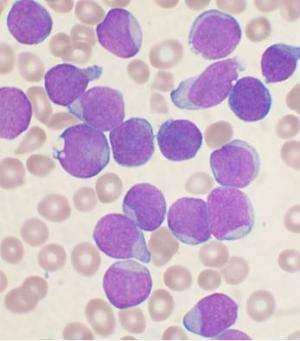
A team in the University of Kent's pharmacy school conducted a study that discovered that leukaemia cells release a protein, known as galctin-9, that prevents a patient's own immune system from killing cancerous blood cells.
Acute Myeloid Leukaemia (AML)—a type of blood cancer that affects over 250,000 people every year worldwide—progresses rapidly because its cells are capable of avoiding the patient's immune surveillance. It does this by inactivating the body's immune cells, cytotoxic T lymphocytes and natural killer (NK) cells.
Existing treatment strategies consist of aggressive chemotherapy and stem cell transplantation, which often do not result in effective remission of the disease. This is because of a lack of understanding of the molecular mechanisms that allow malignant cells to escape attack by the body's immune cells.
Now the researchers at the Medway School of Pharmacy, led by Dr Vadim Sumbayev, Dr Bernhard Gibbs and Professor Yuri Ushkaryov, have found that leukaemia cells—but not healthy blood cells—express a receptor called latrophilin 1 (LPHN1). Stimulation of this receptor causes these cancer cells to release galectin-9, which then prevents the patient's immune system from fighting the cancer cells.
The discovery of this cell mechanism paves the way for new 'biomarkers' for AML diagnosis, as well as potential targets for AML immune therapy, say the researchers.
"Targeting this pathway will crucially enhance patients own immune defences, helping them to eliminate leukaemia cells", said Dr Sumbayev. He added that the discovery has the potential to also be beneficial in the treatment of other cancers.
More information: The research, entitled The Tim-3-galectin-9 Secretory Pathway is Involved in the Immune Escape of Human Acute Myeloid Leukemia Cells (Gonçalves Silva, I., et al.) is published in the journal EBioMedicine. See: www.ebiomedicine.com/article/S … (17)30291-8/fulltext