Chickenpox virus fatal in newly discovered immunodeficiency
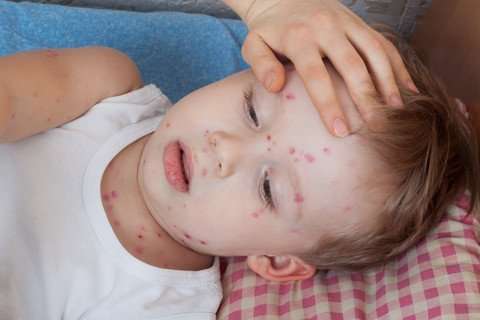
A mutation in one of the sensors that the immune system uses to detect viruses can, in rare cases, turn infections with the chickenpox virus into a life-threatening matter. For two out of every 10,000 people, it can lead to inflammation of the brain, and for twenty out of 10,000, to severe pneumonia, which can be particularly dangerous for pregnant women. The cause of these rare but serious diseases has thus far been unknown, and it has not been possible to predict who was in the danger zone.
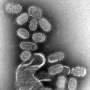
A mapping of the genome in patients who have been very severely affected by these infections has now shown that they have mutations in what is known as the POL III sensor. This is the sensor which the immune system uses to recognise the genome of varicella zoster virus (VZV), which normally simply leads to chicken pox.
The immune system fails to activate
Analyses of cells from the affected patients showed that their cells did not detect the infection and consequently neither did they activate the immune system's defensive responses. This allowed the virus to spread to the brain, for example. If the mutated gene was repaired, the cells were able to fight the infection. This suggests that individuals with mutations in POL III have an increased risk of developing serious illness with the VZV infection.
"We cannot yet put an exact figure on how much the risk of complications is increased when you have this new immunodeficiency, since we have looked at relatively few patients in our study. Neither do we know how large a proportion of all those who have inflammation of the brain and pneumonia have the defect. But we do know that this applies to both children and adults," says one of the driving forces behind the study, Professor with special responsibilities (MSO) Trine Hyrup Mogensen from the Department of Clinical Medicine and the Department of Biomedicine at Aarhus University.
The results have just been published in the scientific journal The Journal of Clinical Investigation.
Paves the way for personalised medicine
The results contribute with a genetic explanation of why some individuals are extremely susceptible to certain infections in contrast to others, and it is an important contribution to an emerging field of research. With the combination of clinical research and basic research, the researchers have succeeded in producing fundamental new knowledge about the immune system, and at the same time, provided a tool that can be used directly in clinical practice.
"Today it is possible to map the entire genomic composition of an individual. At the same time the last few decades have seen an explosion in our knowledge of the immune system. Together this means we are now slowly becoming able to understand the individual differences in susceptibility to infections at both the genetic and molecular level," says Søren Riis Paludan, who is professor at the Department of Biomedicine at Aarhus University and also one of the researchers behind the research project.
"In general terms, it opens up new possibilities with more individualised diagnosis and treatment of patients. Our study is an important contribution to this," adds Trine Hyrup Mogensen, who also works as a medical specialist in infectious diseases at Aarhus University Hospital.
More information: Benson Ogunjimi et al. Inborn errors in RNA polymerase III underlie severe varicella zoster virus infections, Journal of Clinical Investigation (2017). DOI: 10.1172/JCI92280