New tool for cell-free therapy based on artificial membrane vesicles
Scientists at Kazan Federal University's Institute of Fundamental Medicine and Biology, led by Professor Albert Rizvanov, have shown that artificial membrane vesicles generated by Cytochalasin B treatment of human cells retain angiogenic activity.
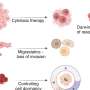
Vesicles are small packages of material released from cells and act to deliver cargo and messages to adjacent and distant cells. Vesicles are known to be important regulators of normal physiology and have also been implicated in disease, notably cancer. Extracellular vesicles exhibit the biological activity of the cell from which they originate. For example, extracellular vesicles of stem cells are able to promote angiogenesis and regeneration. For this reason, extracellular vesicles represent a promising tool for cell-free therapy to deliver biologically active molecules.
However, the yield of naturally occurring vesicles is too low for practical purposes. Recently, several studies demonstrated the ability to generate a large number of membrane vesicles from cultured cells treated with a drug, Cytochalasin B. This cost-effective approach permits the generation of large quantities of extracellular vesicles. However, it remained unknown whether these Cytochalasin B-induced micro-vesicle (CIMVs) retained characteristic biological properties of their parental cells.
To address this, an international team of investigators, led from Kazan Federal University, Russia by Professor Albert Rizvanov, with international collaborators, characterized the biological activity of membrane vesicles.
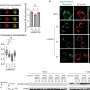
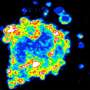
The study was published in Oncotarget. The lead author, Dr. Marina Gomzikova, and colleagues described the morphology, molecular composition, fusion capacity and biological activity of Cytochalasin B-induced membrane vesicles (CIMVs). This data suggests that the biophysical, molecular and size distribution properties of CIMVs are similar to natural vesicles. Furthermore, they demonstrated that CIMVs retain the biological properties of the donor cells, as they can stimulate angiogenesis in vitro and in vivo.
CIMVs can now be produced in large quantities and scaled to an industrial production level; potential therapeutic applications to deliver biologically active molecules of CIMVs are now possible.
More information: Cytochalasin B-induced membrane vesicles convey angiogenic activity of parental cells. Oncotarget. doi.org/10.18632/oncotarget.19723