Strongyloidiasis worm infects many Australians, yet hardly anybody has heard of it

We all know about parasites, like tapeworms that can get into our intestines if we eat infected undercooked meat. There are many types of parasitic worms, including flatworms and roundworms, and they can all make humans sick.
But there's one infection by a parasitic worm that, worldwide, makes more people ill than malaria, and yet hardly anybody has ever heard of it. It's called strongyloidiasis, and estimates suggest up to 60 percent of Indigenous Australians in some communities carry the infection.
What is it?
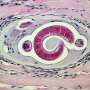
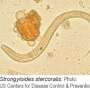
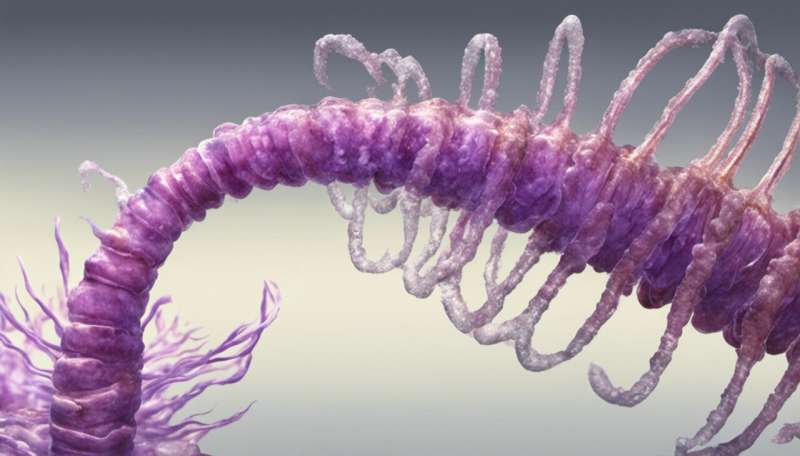
Strongyloidiasis is an infection caused by a parasitic worm called Strongyloides stercoralis (S. stercoralis), which infects the gastrointestinal system (stomach, intestines and other digestive organs). Worldwide, it infects an estimated 370 million people, making it more common than malaria. But so few people are aware of it that it has been described as the most neglected of all neglected diseases.
Strongyloidiasis is generally considered a disease of developing countries, but we also see it in economically disadvantaged areas in the US, Asia and Europe. In Australia, the worm can infect anyone, but is most common in Indigenous Australian communities, refugees, returning overseas travellers and Vietnam veterans.
Estimates suggest the prevalence of infection in Indigenous Australian communities is 35-60%. Yet the true incidence in Australia could be much higher as the infection is difficult to detect, is often not tested for and is not a nationally notifiable disease. This means there is no centralised record of cases.
How can you get infected?
You can be infected with the worm by coming into contact with contaminated soil or faeces. The way it gets into your intestine is quite a process. First it enters your body through skin, normally the feet. From there it makes its way into the bloodstream and then the lungs. It is then coughed up and swallowed, which is how it ends up in the small intestine. It can live there for decades.
Infection can be undetected for many years. Patients can show no symptoms or suffer vague ones like weight loss, indigestion, abdominal cramps, diarrhoea, coughing, wheezing or a rash like hives on the buttocks or waist. Diagnosis can be confirmed by detecting the worm in a stool sample or looking for antibodies (which means the immune system has developed proteins to fight the infection) in a blood sample.
However, the detection methods are not foolproof. Stool sample analysis can result in false negatives because the worms can move into the faeces at different times. And blood tests can result in false positives due to the detection method reacting to other proteins and mistaking them for Strongyloides.
If infection goes undiagnosed and your immune system becomes compromised, or you are given steroids which suppress the immune response, the parasite can enter new phases of infection. These are hyperinfection or dissemination.
Hyperinfection is when the worm reproduces rapidly. The result is an enormous number of worms, which is often deadly.
Dissemination is when the worms spread throughout the body. This is almost always fatal as the gut bacteria carried by the worm are moved into other organs, causing massive infection.
How is it treated?
If diagnosed correctly, the infection can be treated by a drug called ivermectin, which is typically used against parasitic worms and other parasites including scabies. However, if one worm remains, it can reproduce (asexually), causing reinfection. Also, you do not develop immunity to the worm, so you can be immediately reinfected once the drug has left your system.
The biggest issue with drug treatment is that we have already started to see ivermectin-resistant Strongyloides in sheep and horses. Another drug family, called benzimidazoles, is sometimes used against human Strongyloides, but we've also seen resistance to this in infected animals.
Mass drug administration of ivermectin has been successful in significantly reducing the number of infected people in some communities in Australia. However, its roll-out has been patchy. Also reinfection can occur, suggesting the environmental part of the Strongyloides lifecycle plays an important role.
Controlling the environment
Very little is known about how the worm survives outside a host and little has been done to try to control it in the environment. The worm can reproduce once outside a host, but we don't know how long it can survive in the environment, although it is thought to be quite a while.
The worm's survival in the environment explains the prevalence of strongyloidiasis in low socioeconomic areas. Poor infrastructure, living conditions and sanitation result in contaminated soil and water where the worm can live and cause infection.
We must start targeting Strongyloides control in the environment and reduce our reliance on drugs. If human Strongyloides becomes resistant to ivermectin, the consequences could be devastating. However, the biggest challenge in fighting this disease is that many people have not even heard of the Strongyloides worm.
This article was originally published on The Conversation. Read the original article.![]()