New drug reduces rate of progression of incurable eye disease
An international study including researchers from the Centre for Eye Research Australia (CERA) has found a way to slow the progression of dry age-related macular degeneration (AMD) - one of the most common causes of vision loss in people over the age of 50.
The Phase 2 clinical trial (known as the FILLY trial) was sponsored by Apellis Pharmaceuticals and included 246 patients across 40 testing sites, seven of which were in Australia.
Principal Investigator Robyn Guymer from CERA and the University of Melbourne says she is delighted with the results.
"In the past decade, we have made tremendous advances in treating one of the late complications of AMD which is called wet AMD, where blood vessels leak in the back of the eye and destroy vision rapidly," Professor Guymer says.
"Now we are directing our attention to treating the other irreversible late complication of AMD, called dry AMD or geographic atrophy (GA)."
Dry AMD or GA is a condition where the cells in the retina die slowly over many years, eventually leading to irreversible loss of vision.
"It is like having moth eaten holes in your vision and they slowly all join up in the middle part of the vision, destroying the ability to read, drive and recognizing faces," Professor Guymer says.
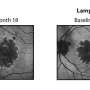
Apellis Pharmaceuticals developed a new compound called APL-2 for treating these patients with GA. Patients were given injections either monthly or every other month for 12 months, resulting in a reduction in GA lesion growth of 29 per cent and 20 per cent respectively, compared to control patients.
Additionally, in a post hoc analysis, a greater effect was observed during the second six months of the study: a reduction in GA lesion growth rate of 47 per cent with monthly administration, and a reduction of 33 per cent with every other month administration.
Based on these positive results, Apellis plans to proceed with Phase 3 studies as soon as possible.
The FILLY trial is a 246-patient Phase 2 multicenter, randomized, single-masked, sham-controlled clinical trial of APL-2 in patients with GA conducted at 40 clinical sites, located in the United States, Australia and New Zealand. APL-2 was administered as an intravitreal injection in the study eye monthly or every other month for 12 months, followed by six months of monitoring after the end of treatment. Eyes were evaluated for GA by fundus autofluorescence photographs (FAF). The rate of GA area growth was measured by mean change in square root area of GA lesion from baseline to month 12. The primary endpoint was the change in GA lesion size from baseline to month 12, compared to sham.
GA is an advanced form of age-related macular degeneration (AMD), a disorder of the central portion of the retina, known as the macula, which is responsible for central vision and colour perception. GA is a chronic, progressive condition that leads to central blind spots and permanent loss of vision. There are currently no approved treatments for GA.