A new strategy induces the regression of advanced lung tumors in mice
A study published in Cancer Cell by researchers of the Molecular Oncology Program at the Spanish National Cancer Research Centre (CNIO) shows that the elimination of the c-Raf kinase by genetic manipulation causes the regression of Kras oncogene-driven advanced lung tumours in a genetically designed mouse model that reproduces the natural history of this tumour type. It also demonstrates that the elimination of the c-Raf protein produces tolerable toxic effects. This opens a new possibility for the development of therapies against tumours for which there are still no selective medicines and which therefore must be treated with cytotoxic drugs with abundant side effects.
One in four human solid tumours harbours mutations in KRAS with very poor prognosis, such as adenocarcinomas of the pancreas, lung and colon. The alteration of this gene directly and indirectly affects cell proliferation and differentiation through the activation of multiple signalling pathways, key phenomena in tumour development. However, there are no approved compounds in the clinic that selectively attack these pathways present in these carcinomas.
Blocking these oncogenic signaling pathways without affecting normal homeostasis is "one of the greatest challenges of precision medicine," the authors note. In the case of KRAS signalling, attempts made to date with the inhibition of the MEK and ERK kinases, although effective in preventing the development of the tumour, induce unacceptable toxicity.
Lung cancer is the leading cause of cancer-related deaths worldwide, accounting for an estimated 1.8 million new cases per year. Non-small-cell lung carcinoma (NSCLC), which is the most frequent subtype of lung cancer, also carries mutations in KRAS in up to 30 percent of cases. Additionally, the 5-year survival rate for lung cancer remains below 15 percent, thus underscoring the urgent need to improve the current treatment for these cancer patients.
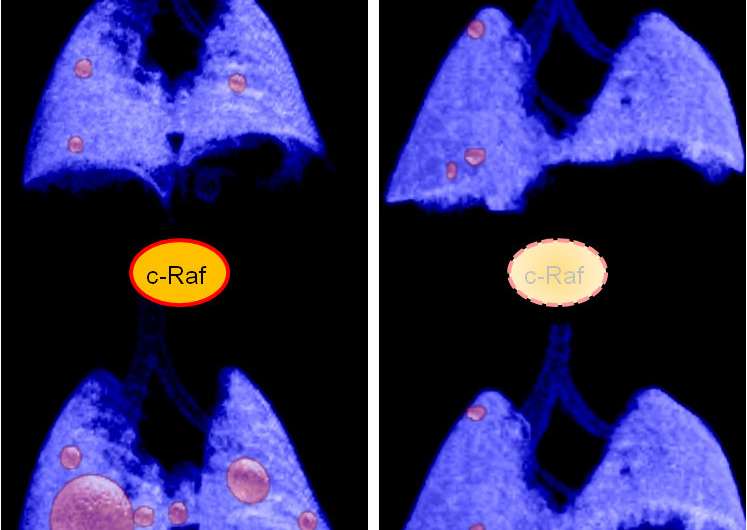
In this new work, researchers have genetically engineered a new mouse model eliminating c-Raf not only in advanced lung tumours but systemically throughout the body of the animal. This could allow clinicians to predict the possible toxicity of drugs that block the activity of c-Raf.
Monica Musteanu, one of the work's main authors, says the study shows that the elimination of c-Raf "provides a significant therapeutic benefit that results in the regression of most tumours with very acceptable levels of toxicity." However, the authors warn that in this study, the therapeutic effect requires the elimination of the protein, something that at the moment is not possible to achieve through pharmacological strategies. Since c-Raf is a protein kinase, in principle, the results described in this study could be validated by using selective inhibitors of this enzyme's activity. In fact, the main priority of the authors is now to determine to what extent the therapeutic activity depends on c-Raf kinase activity. The results that derive from these ongoing studies will serve to define what pharmacological strategy may be applied in the future to treat cancer patients.
More information: Cancer Cell (2018). DOI: 10.1016/j.ccell.2017.12.014