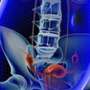
Ulipristal achieves amenorrhea for symptomatic leiomyomas
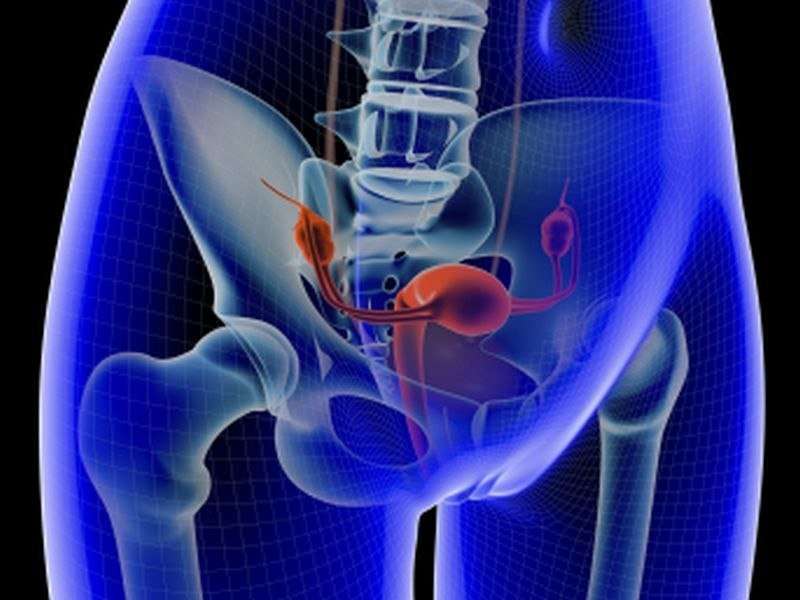
(HealthDay)—For women with symptomatic uterine leiomyomas, ulipristal is well tolerated and superior to placebo for the rate of and time to amenorrhea, according to a study published online Feb. 5 in Obstetrics & Gynecology.
James A. Simon, M.D., from the George Washington University School of Medicine in Washington, D.C., and colleagues enrolled premenopausal women with abnormal uterine bleeding, one or more discrete leiomyomas, and uterine size 20 weeks gestation or less. One hundred fifty-seven patients were randomly allocated to receive 5 mg ulipristal, 10 mg ulipristal, or placebo (53, 48, and 56 patients, respectively) for 12 weeks followed by 12-week drug-free follow-up.
The researchers found that amenorrhea was achieved by 47.2, 58.3, and 1.8 percent of patients in the ulipristal 5 mg and 10 mg, and placebo groups, respectively. For both ulipristal doses, the time to amenorrhea was shorter than placebo; compared with placebo, both ulipristal doses resulted in improved quality of life. Hypertension, elevated blood creatinine phosphokinase, and hot flushes were common adverse events (5 percent of greater in either ulipristal group during treatment). Four patients experienced serious adverse events, but none was considered related to treatment.
"Ulipristal at 5 mg and 10 mg were well tolerated and superior to placebo in rate of and time to amenorrhea in women with symptomatic uterine leiomyomas," the authors write.
Several authors disclosed financial ties to pharmaceutical companies, including Allergan, which funded the study.
More information: Abstract/Full Text (subscription or payment may be required)
Copyright © 2018 HealthDay. All rights reserved.