Uterine lining test improves chances of a successful pregnancy

It takes a healthy embryo and a healthy mother to nurture the microscopic meeting of a sperm and an egg through nine months of development before a baby can be born.
Even as four million babies are born in the United States every year, about 6 percent of married women of reproductive age in the United States cannot become pregnant after a year of trying. And about 25 percent of all women who become pregnant have a first-trimester loss.
There are many causes of infertility, including secondary effects of various disorders as well as hormonal and genetic conditions in either the man or woman. And many factors can influence how or if an embryo grows into a healthy breathing baby.
One factor that Dr. Harvey Kliman has studied for nearly three decades is the function of the endometrium, the mucous membrane that lines the inside of the uterus and serves as the nutrient-rich medium into which an embryo must implant before it can grow.
"You can have the perfect embryo, but if you don't have perfect medium, it won't grow," Kliman said, noting how the endometrium feeds the embryo until the ninth week of pregnancy, when the mother's blood takes over through the embryo's placenta.
Kliman serves as Director of the Reproductive and Placental Research Unit in Yale School of Medicine's Department of Obstetrics, Gynecology and Reproductive Sciences. He began exploring a way to test the ability of the endometrium to support a pregnancy shortly before arriving at Yale from the University of Pennsylvania in 1991, hoping to find a clue within the endometrium's constantly changing nature.
"Various organs within our bodies, such as the liver and kidneys, are constant," he said. "They are basically the same, day in and day out. But the endometrium is unique. It's the only organ that changes over 28 days."
Over the course of a normal menstrual cycle, the endometrium alters its structure every day under the influence of fluctuating hormones in preparation for the possibility it will be needed to support a fertilized egg. The lining slowly builds up and then sheds, passing through the vagina as menstrual fluid before the process starts again.
"The endometrium then needs to get to a second phase, when it is no longer growing, before the embryo arrives," Kliman said. "If this process stops prematurely, it won't support implantation."
The Search for a Telltale Clue
In 1998, newly founded Women's Health Research at Yale awarded Kliman one of the center's first Pilot Project Program grants to study whether the presence or absence of a particular mucin—a protein with many long sugar groups—at a certain stage of the menstrual cycle could serve as an indicator of healthy endometrial function and better chances for a successful pregnancy.
The mucin, named MAG, creates long filaments that the endometrium sends out to slow down a newly created embryo approaching from a Fallopian tube.
"It's like sending lines from a dock to a boat floating downstream with help from workers on the shore," Kliman said. "The embryo is the boat, and the mother is the shore."
About 90 percent of embryos don't attach, mostly due to genetic problems with the embryo, Kliman said. But for
some women, the endometrium doesn't work properly to allow implantation.
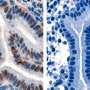
Kliman noted that MAG is only present in the first phase of the menstrual cycle, when the endometrium is growing. Using the WHRY grant, Kliman studied biopsies obtained from women with and without fertility problems and found that 85 percent of fertile women displayed normal patterns of MAG. But only 20 percent of women with infertility issues showed normal MAG patterns. Almost 30 percent of women with fertility problems had none of this mucin at all.
"This was a significant step," Kliman said. "That first study gave us the framework to look for something that turned on and off at different phases of the menstrual cycle. It showed the potential pathway."
But MAG is only present in women with Type A or AB blood, roughly 45 percent of the population.
"If we are using an instrument to shine a light into the unknown, MAG is very limited," Kliman said. "We realized it wasn't the solution we needed."
An Even Better Test
Armed with this new knowledge and now adept at obtaining biopsy samples from women with different degrees of fertility, Kliman and his colleagues shifted their attention. And their thinking.
Instead of looking at one or more markers produced by the endometrium to facilitate implantation, Kliman began to examine something fundamental to all cell growth.
Whenever a cell divides to create new cells, it must bypass biological checkpoints that regulate whether the process continues or stops. Cancer cells, for example, open all the checkpoints so they can continue to divide and spread uncontrolled.
Cyclin is a type of protein that forms one of these checkpoints to control the cell cycle and determine how cells spread. In the menstrual cycle, a cyclin known as Cyclin E allows the creation of DNA that guides the first phase buildup of the uterine lining. In the second half of the cycle, a gene known as p27 creates a protein that inhibits cell division and stops endometrial growth.
"This provides a much simpler, much more reliable, more uniform way to determine endometrial function," Kliman said. "And it is present in every patient."
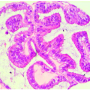
Kliman and his colleagues published their findings in 2003 and soon trademarked and patented their Endometrial Function Test (EFT). His lab has found that women with an abnormal EFT are 10 times less likely to become pregnant than a woman with normal results. But after successful treatment, the women with abnormal EFTs show the same success rates as the general population.
The most common cause of an abnormal EFT is endometriosis, a condition in which the endometrial tissue grows outside the uterus and has no way of leaving the body after it thickens and breaks down during menstruation. After medical or surgical treatment, women with endometriosis are more likely to carry a pregnancy to term, Kliman said.
Other problems leading to an abnormal EFT include obesity or extreme thinness, which is often a sign of excessive exercise and high levels of stress that can make the body inhospitable for an embryo. Anatomic problems might include something called hydrosalpinx, in which fluid builds up in the Fallopian tubes and backs up into the uterus.
But Kliman cautions women not to consider the EFT a magic pill to cure their infertility, which can be caused by problems involving the sperm as well as problems with functions in organs such as the brain, pituitary gland, and ovaries. He said that the process and his recommendations are complex and should be addressed by experts who are board certified in reproductive endocrinology and infertility.
Still, Kliman sees his test as a way to address the emotionally devastating clinical problem of infertility for women who wish to have children and to effectively guide women to the best fertility treatment plan for them, possibly avoiding the expensive effort of in vitro fertilization (IVF).
"Now, the most common way to test for endometrial function is to see how a pregnancy develops," Kliman said. "But an embryo is so important, we should first make sure the endometrium is receptive. Because we want to implant that embryo in an environment that gives it the best opportunity to grow."