A neuron can cause a domino effect

Loss of the sense of smell can indicate a neural disease like Alzheimer's or Parkinson's disease. However, contrary to previous belief, degenerations in the nervous system do not play a leading role in the loss of the sense of smell with increasing age. Rather, individual nerve cells or classes of nerves are responsible.
Some nerve cells (neurons) or neuron classes in the brain seem to age faster than others. For example, the loss of the olfactory sense is one of the first clinical signs of natural aging. This can be accompanied by a neurodegenerative disease such as Alzheimer's.
"Age is the major risk factor for Alzheimer's or Parkinson's disease," says Prof. Ilona Grunwald Kadow from the School of Life Sciences at the Technical University of Munich (TUM). "Only a small proportion of these diseases are due to known genetic reasons". The question is why do some neurons age faster than others. Why are some more sensitive? And is the damage to certain types of neurons the reason why entire nerve networks fail to function properly?
A new study conducted under the direction of Prof. Grunwald Kadow now shows how the olfactory capacity of the fruit fly ages, and how much this resembles the aging process in the human olfactory system. Like humans, the fruit fly loses its powers of smell as it ages. Several key genes and mechanisms were identified that contribute to this aging-associated degeneration.
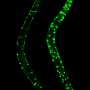
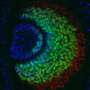
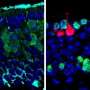
In the next step, the scientists examined whether all or only specific neurons of the olfactory circuit were affected. The team found that some neurons are more sensitive than others, and decline faster during aging. They determined that oxidative stress alters specific neuron types, causing the gradual collapse of neural network function. Oxidative stress causes the accumulation of reactive oxygen compounds in the cells or tissue, which can cause temporary or permanent damage and accelerated aging.
Interestingly, the prevention of these reactive oxygen compounds in only this type of neuron completely stopped the loss of sense of smell. Old flies sensed odors as well as their young conspecifics. This suggests that age-related degeneration could be significantly delayed by preventing oxidative damage in only one or a few neuron types.
A trial with the antioxidant resveratrol in younger flies over several weeks showed that it can counteract oxidative stress, which develops during aging. This treatment appeared to protect particularly sensitive neurons and thereby contributed to maintaining their function within the neural network. In the elderly, such treatments might help to delay the onset of neurodegenerative diseases associated with aging.
Another possible factor that could play a role in the aging process is the intestinal microbiome. It could be involved in the progression of Parkinson's disease. Grunwald Kadow and her team have therefore also tested the effect of specific microbiota on olfactory aging in fruit flies with the result that certain bacteria have a positive effect and slow down olfactory neurodegeneration.
Prof. Grunwald Kadow says these findings and ongoing experiments in the fruit fly model could pave the way for more targeted treatments and therapies.
More information: Ashiq Hussain et al, Inhibition of oxidative stress in cholinergic projection neurons fully rescues aging-associated olfactory circuit degeneration in Drosophila, eLife (2018). DOI: 10.7554/eLife.32018