First real-world studies report glecaprevir/pibrentasvir to be effective and well tolerated in chronic HCV infection
The results of the first real-world studies assessing the effectiveness and safety of glecaprevir/pibrentasvir (G/P) in patients with chronic hepatitis C virus (HCV) infection have confirmed high rates of viral suppression and a favourable safety profile in patients receiving 8-16 weeks of treatment. Two real-world studies from Italy and Germany which will be presented at this week's International Liver Congress 2018 in Paris, France, reported high rates of sustained virological response (SVR), defined as undetectable HCV RNA, at 4 and 12 weeks after the end of treatment.
"The efficacy and safety of G/P as a treatment for HCV-infected patients have so far only been evaluated in controlled clinical trials," explained Dr. Roberta D"Ambrosio from the University of Milan in Italy. "Our real-world study involving more than 700 patients with chronic HCV infection confirmed that the effectiveness and safety profile of G/P were excellent across a range of different patient types."
Glecaprevir (an NS3/4A protease inhibitor) coformulated with pibrentasvir (an NS5A inhibitor) is a relatively new direct-acting antiviral (DAA) combination that was approved in multiple countries during 2017 for the treatment of chronic HCV infection in adults.1 Phase 2 and 3 studies involving tightly defined patient groups with HCV infection have reported high rates of SVR12 and a favourable safety profile.1-6 Until now, no real-world studies with G/P in broader groups of patients with HCV infection have been reported.
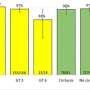
The Italian study being presented this week is an interim analysis evaluating the outcomes of 723 consecutively treated patients within the Lombardy Navigator-II Network, with G/P administered according to the drug label. Of those with available data, 99.7% achieved SVR4 (346/347). HCV RNA was reported to be undetectable in 74% of patients at Week 4, and in 98% of patients at end of treatment for the entire cohort. The prevalence of treatment-related adverse events was low, mainly of mild severity, and only three patients discontinued G/P treatment prematurely.
The ongoing German real-world study, also being reported this week, evaluated 638 patients from the German Hepatitis C-Registry (DHC-R) who received G/P treatment according to the local label. Adult patients with HCV genotypes 1-6, with or without compensated cirrhosis, who were either treatment-naïve or treatment-experienced were included in this interim analysis. The majority of patients were treatment-naïve without cirrhosis and treated with 8 weeks of G/P.
According to Prof. Dr. Thomas Berg from the University of Leipzig in Germany, who will present the study findings in Paris, among the 49 patients with available data, 100% achieved SVR12, excluding four patients who prematurely discontinued treatment for reasons other than virological failure. Of those four patients, two discontinued treatment due to adverse events. No grade 3 or higher elevations in alanine aminotransferase (ALT) have been observed.
"Our real-world study in patients receiving G/P in everyday clinical practice has yielded favourable effectiveness and safety results that were consistent with the clinical trial data," said Prof. Dr. Thomas Berg. "We have found G/P to be a very useful addition to our HCV treatment armamentarium as it simplifies treatment decisions for the majority of patients; G/P has the potential to expand the treated population and support the goal of HCV elimination."
"These data are important because they confirm the high cure rates of more than 98% observed in Phase 3 trials," said Prof. Markus Cornberg from the Hannover Medical School, Germany, and EASL Governing Board Member. '8 weeks of therapy is possible for all naïve, non-cirrhotic patients, regardless of genotype, and although we still lack data in some difficult-to-treat genotype 3 patients, prevalence of these seems to be declining as shown by the German registry."
More information: References:
1. AbbVie Limited. Maviret Summary of Product Characteristics, 28 February 2018. Available from: www.ema.europa.eu/docs/en_GB/d … 4430/WC500233677.pdf. Last accessed: March 2018.
2. Zeuzem S, et al. Glecaprevir-pibrentasvir for 8 or 12 weeks in HCV genotype 1 or 3 infection. N Engl J Med. 2018;378(4):354-69.
3. Gane E, et al. Glecaprevir and pibrentasvir in patients with HCV and severe renal impairment. N Engl J Med. 2017;377(15):1448-55.
4. Forns X, et al. Glecaprevir plus pibrentasvir for chronic hepatitis C virus genotype 1, 2, 4, 5, or 6 infection in adults with compensated cirrhosis (EXPEDITION-1): a single-arm, open-label, multicentre phase 3 trial. Lancet Infect Dis. 2017;17(10):1062-8.
5. Asselah T, et al. Efficacy of glecaprevir/pibrentasvir for 8 or 12 weeks in patients with hepatitis C virus genotype 2, 4, 5, or 6 infection without cirrhosis. Clin Gastroenterol Hepatol. 2018;16(3):417-26.
6. Kwo PY, et al. Glecaprevir and pibrentasvir yield high response rates in patients with HCV genotype 1-6 without cirrhosis. J Hepatol. 2017;67(2):263-71.