Dicer enzyme cuts down on fats
The enzyme Dicer cleaves long precursors into short RNA molecules called microRNAs. A new study reveals how Dicer enhances energy metabolism and reduces levels of fat storage in macrophages, thus slowing the progression of atherosclerosis.
Atherosclerosis is one of the primary causes of premature death in modern societies. The condition is characterized by the deposition of fat-soluble molecules – principally cholesterol and triglycerides – on the inner walls of major blood vessels. This process triggers vascular inflammation and the formation of atherosclerotic plaques, which ultimately lead to narrowing of the arteries, thus impeding the flow of blood. Cells called macrophages are responsible for the uptake and disposal of the fatty deposits that induce plaque formation. However, depending on how they have been activated, macrophages can play an insidious role in the development of atherosclerosis. Inflammatory activation promotes inflammation, while 'alternatively activated' macrophages suppress inflammation reactions. The latter degrade triglycerides by means of fatty acid oxidation, but if this pathway operates suboptimally – due to local inflammation, for example – they accumulate the engulfed fats, thus contributing to plaque development and increasing the risk of blood vessel blockage. A team of researchers in the Institute for Prophylaxis and Epidemiology of Cardiovascular Diseases at the LMU Medical Center, led by Professor Andreas Schober, has now shown that the enzyme Dicer plays an important role in the breakdown of triglycerides in alternatively activated macrophages. The findings appear in the latest issue of the journal Circulation.
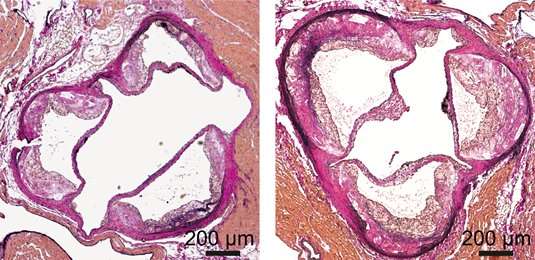
Dicer slices long precursor RNAs into short snippets, which are known as microRNAs (miRNAs). miRNAs make a significant contribution to the regulation of gene activity. By binding to complementary nucleotide sequences in messenger RNAs (mRNAs), miRNAs prevent the synthesis of specific proteins. The Munich researchers used a mouse model system to study the impact of functional deletion of the Dicer gene in macrophages on energy metabolism. "We found that lack of Dicer in macrophages impairs their capacity for oxidative degradation of triglycerides. As a result, they accumulate more fat and develop into what are called foam cells", Schober explains. Foam cells die at a higher rate than normal macrophages, which stimulates progression of atherosclerosis.
Schober and his colleagues went on to show that the effects of Dicer knockout on macrophage function are attributable to the loss of a single microRNA, named miR-10a, which is required for normal regulation of energy metabolism in these cells, and boosts the oxidative breakdown of the fatty acids in triglycerides. In addition, they identified the relevant target of miR-10a as the mRNA that codes for the ligand-dependent nuclear co-repressor protein (Lcor). "When the interaction between the two RNAs (which reduces levels of the Lcor protein) is inhibited, progression of atherosclerosis is accelerated, as in the case of the knockout of Dicer," says Schober.
The discovery of this specific regulation of fatty acid metabolism in macrophages could lead to a novel therapeutic strategy for the treatment of atherosclerosis. Schober and his team now plan to explore ways of suppressing the negative effects of inflammation on fatty acid oxidation in alternatively activated macrophages.
More information: Yuanyuan Wei et al. Dicer in Macrophages Prevents Atherosclerosis by Promoting Mitochondrial Oxidative Metabolism, Circulation (2018). DOI: 10.1161/CIRCULATIONAHA.117.031589