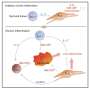
Immune response associated with inflammation and joint damage in rheumatoid arthritis
To diagnose rheumatoid arthritis, antibodies to the amino acid citrulline are commonly measured. A new study from Uppsala University shows that a broad mix of different antibodies in the joints is the dominant factor that can be associated with severe inflammation and joint damage. These findings, published in Annals of the Rheumatic Diseases, may eventually lead to improved diagnostics.
Rheumatoid arthritis (RA) is a chronic inflammatory disease that affects joints and other organs. To assist diagnosis of the disease, analysis of antibodies to the amino acid citrulline is performed. These ACPAs (anti-citrullinated protein antibodies), which form in response to inflammation, were first described some 20 years ago. ACPAs can be demonstrated in roughly two-thirds of all patients with rheumatoid arthritis.
Subsequent research has shown genetic differences between RA patients with and without ACPAs. The onset of ACPA-positive RA has also proved to be strongly associated with smoking, while this does not apply to RA patients without ACPA.
"So RA patients with and without ACPA probably have different diseases with separate causes, but start with a similar clinical picture," explains Professor Johan Rönnelid, who heads the research team behind the study.
As a category, RA patients with ACPAs have more joint inflammation, with more joint damage assessed by means of X-rays, than RA patients without ACPAs. Common ACPA tests are designed to detect most ACPAs irrespective of which proteins they react with, but it is known that different RA patients react by producing antibodies against different citrullinated proteins or protein fragments.
When auto-antibodies bind to corresponding substances naturally produced by (native to) the body, immune complexes are formed that can then trigger inflammation. As two research groups have shown, ACPAs can form these immune complexes in vitro and cause inflammation there. ACPAs have been assumed to induce inflammation through their presence in immune complexes in the inflamed joint.
"But no one has previously shown that immune complexes containing ACPAs are present in RA patients' body fluids. Overall, there has been very little research on which antibodies are present in immune complexes associated with different diseases, since practicable lab methods have been lacking," Rönnelid says.
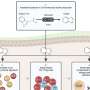
Azita Sohrabian, a Ph.D. student and research engineer at the Department of Immunology, Genetics and Pathology at Uppsala University, has developed a method of isolating immune complexes from various body fluids, and then combined this method with measurement of 19 ACPAs against various citrulline-containing protein fragments in serum and synovial fluid, and in immune complexes, from 77 RA patients. No single ACPA was found to be generally more associated than the other ACPAs with the degree of inflammation and joint damage. On the other hand, the number of different ACPAs that it was possible to demonstrate in the immune complexes from inflamed joints was associated with several different measures of joint destruction and inflammation.
These findings support the suspicion that ACPAs can be pathogenic by forming immune complexes in the joints of RA patients, where these immune complexes can then cause joint inflammation. No single ACPA appears to be particularly important. Instead, the results indicate that a wide range of ACPAs in immune complexes from the joints can induce the local inflammation and drive the process of joint damage in RA. These findings correspond to what has previously been shown in antibody-induced joint inflammation in experimental animals, where various different antibodies to the same protein are required simultaneously to generate inflammation.
More information: Sohrabian A, Mathsson-Alm L, Hansson M, et al. Ann Rheum Dis, Epub ahead of print 12062018, DOI: 10.1136/annrheumdis-2017-212627