The four survival strategies of tumor cells in childhood cancer
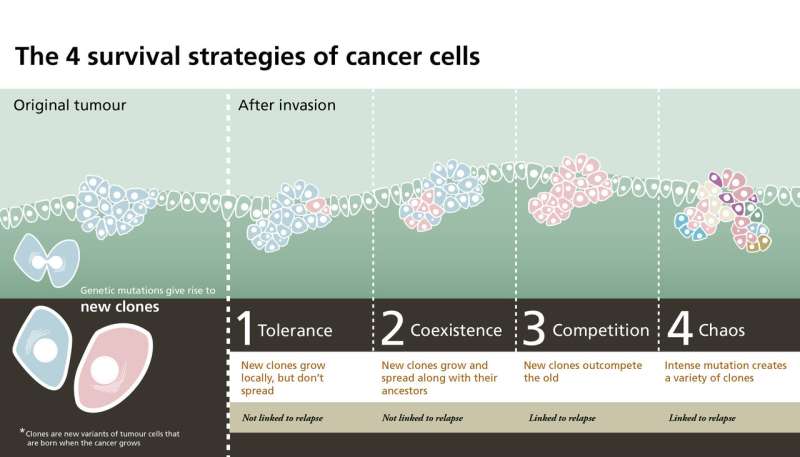
Cancer cells in children tend to develop by following four main trajectories—and two of them are linked to relapse of the disease, according to a study led by Lund University in Sweden, now published in Nature Genetics. The four strategies can occur simultaneously in a single tumour.
The researchers mapped out the genomes of cancer cells from more than 50 tumours in order to identify the four strategies. The genomes of cancer cells often evolve, both to avoid the body's own defence mechanisms and to survive treatment with chemotherapy or other drugs. When cancer cells multiply, mutations develop, and new types of tumour cells, known as clones, can occur.
A challenge when treating patients is that within a single tumour, there may be several distinct clones, which individually trigger the development of cancer in varying ways. The clones may also respond to chemotherapy differently. More knowledge about how such clones develop is therefore an important part of improving treatment.
"We wanted to learn more about how some tumours evade treatment and the strategies the cancer cells develop," explains Jenny Karlsson at Lund University, one of the researchers behind the study.
The developmental trajectories of tumours have so far been unknown in childhood cancer. Therefore, the researchers mapped out the genomes of cancer cells from more than 50 tumours from patients with Wilms tumour, neuroblastoma and rhabdomyosarcoma. This allowed the researchers to track the types of mutations that caused the emergence of four main survival strategies: tolerance, coexistence, competition and chaos.
"The strategies are key, as they give us an indication of the evolutionary capacity a tumour has at the time of discovery. Patients with the first two variants generally have good outcomes, while the latter two strategies are associated with risk of relapse," says Professor David Gisselsson Nord, who led the study.
If two of the strategies—competition or chaos—exist in the tumour at the onset of illness, the risk of relapse is more than 50 percent.
"The same two strategies were found when we analysed relapse tumours. It seems that some cancer cells are programmed from the outset to single-handedly create a relapse. Relapsed tumours had genomes that were often radically altered compared to the patient's first tumour. We conclude that the first tumour should not be used as a proxy to predict targeted treatment in case of a relapse. A new biopsy is well warranted. The genome of the tumour usually changes over time," says David Gisselsson Nord.
The next step will be to identify which mechanisms drive the survival strategies adopted by the cancer in the initial phase of the disease.
"If we knew more about how the environment in the patient's tissues triggers cancer cells to develop, we could also influence how they change during treatment and perhaps prevent a relapse. We are now applying for funding to conduct such studies and to evaluate, in a major study, whether the four strategies can really be used in the clinic," concludes David Gisselsson Nord.
The four strategies:
- Tolerance. New clones are allowed to emerge locally in the tumour, but they stay in their place of origin. The researchers did not find that this strategy was associated to relapse.
- Coexistence. New clones grow together with the original tumour cells, and coexist with them in many parts of the tumour. The researchers did not find that this strategy was associated to relapse.
- Competition. A new clone outcompetes the original tumour cell, and then builds up parts of the tumour entirely on its own. The researchers found that this strategy was associated with increased risk of relapse.
- Chaos. New clones mutate intensively so that a variety of cell types emerge in a specific part of the tumour at the same time. This strategy was also associated with increased risk of relapse.
More information: Jenny Karlsson et al. Four evolutionary trajectories underlie genetic intratumoral variation in childhood cancer, Nature Genetics (2018). DOI: 10.1038/s41588-018-0131-y