FDA drops Zika testing for blood donors
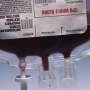
(HealthDay)—Individual blood donations will no longer need to be tested for the Zika virus, according to the U.S. Food and Drug Administration.
"When Zika virus first emerged, the unknown course of the epidemic and the observed severe effects from the disease indicated that individual donor testing was needed to ensure the continued safety of the blood supply," Peter Marks, M.D., Ph.D., director of the FDA's Center for Biologics Evaluation and Research, explained in an agency press release. "Now, given the significant decrease in cases of Zika virus infection in the United States and its territories, we are moving away from testing each individual donation to testing pooled donations."
Pooled testing of donations is considered less burdensome and more cost-effective for blood establishments. However, the FDA will continue to monitor the situation closely, and if necessary, reconsider what measures are needed to maintain the safety of the blood supply.
More information: More Information
Copyright © 2018 HealthDay. All rights reserved.