New high-throughput screening study may open up for future Parkinson's disease therapy
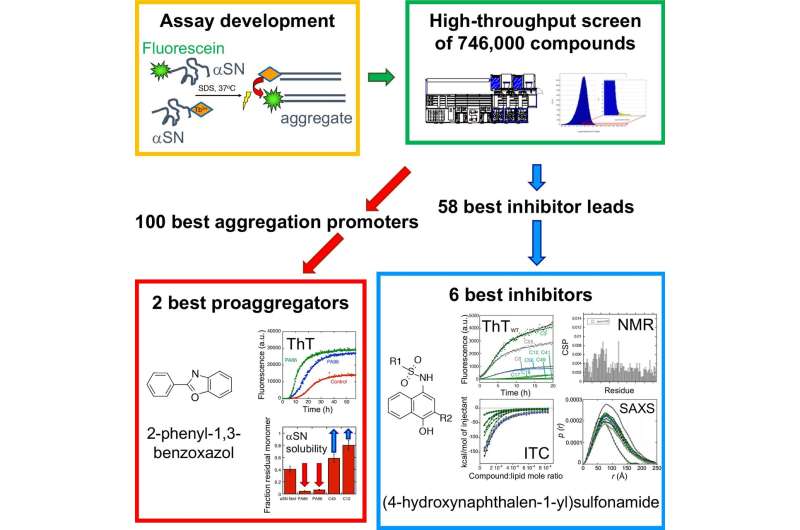
Parkinson's disease (PD) is the most common movement disorder in the world. PD patients suffer from shaking, rigidity, slowness of movement and difficulty with walking. It is a neurodegenerative disease caused by the loss of dopaminergic neurons in the brain. Currently, PD cannot be cured or even halted, but symptoms may be treated to some degree. Probably the single most important cause of PD is the aggregation of the natively unfolded protein α-synuclein (αSN). αSN can form both small oligomeric complexes (αSOs) as well as large fibrillary deposits; the αSOs are thought to be the most toxic species. Preventing or reducing αSN aggregation could be a good way to halt PD development. So far, it has been difficult screen large numbers of compounds to identify potential aggregation inhibitors, since αSN aggregates in a rather irregular and variable fashion; it is also difficult to detect early-stage αSOs.
However, in the new screening strategy, the researchers first developed a smart trick to make αSN aggregate in a more predictable way using the "soap" molecule sodium dodecyl sulfate. To detect the aggregates, they used Förster resonance energy transfer (FRET), a widely used technique for measuring distances within and between molecules. In this way, they were able to screen 746,000 compounds for their ability to inhibit αSN aggregation.
By sifting through the results, they came up with a collection of novel, structurally diverse small compounds that either prevent or accelerate αSN aggregation. The six best inhibitors share a common core structure, and these compounds all interact with the first part of αSN, called the N-terminal region.
The results are exciting in two ways. First, the identified inhibitory molecules could be useful starting points to develop therapy against PD. Second, the compounds can also be used to find out more about how αSN aggregation in the cell affects PD development and thus understand more about the molecular basis for PD.
More information: Martin Kurnik et al, Potent α-Synuclein Aggregation Inhibitors, Identified by High-Throughput Screening, Mainly Target the Monomeric State, Cell Chemical Biology (2018). DOI: 10.1016/j.chembiol.2018.08.005


















