Bacterial imaging probe is safe for patient use, study finds
Imaging technology that detects deadly pneumonia infections in under 60 seconds is safe and practical for clinical use, a study has found.
Tests on patients with suspected infections found the approach could detect bacteria deep inside the lung where other technologies fail to reach.
Experts say the technology will enable bedside decision making in critically ill patients, helping to avoid unnecessary use of antibiotics.
It could also help doctors to better monitor patients' illness, so that treatment can be stopped once infection has cleared, or if alternative therapies are needed.
The technology could also help us to better understand lung infections, paving the way to develop improved treatments.
Each year, across the world, around 20 million patients in intensive care need ventilators to help them breathe. Up to one third are suspected to have serious lung infections, such as pneumonia. This leads to huge antibiotic use, which can increase drug resistance and make bacteria harder to treat.
Doctors currently test fluid samples from patients' lungs to check if bacteria are present, but these tests can take days to return a result and may be inaccurate.
They can also use x-rays, which are overly sensitive and can lead to patients being treated with powerful but unnecessary antibiotics.
Monitoring patient responses to these antibiotic treatments in real time is not feasible with either of these approaches.
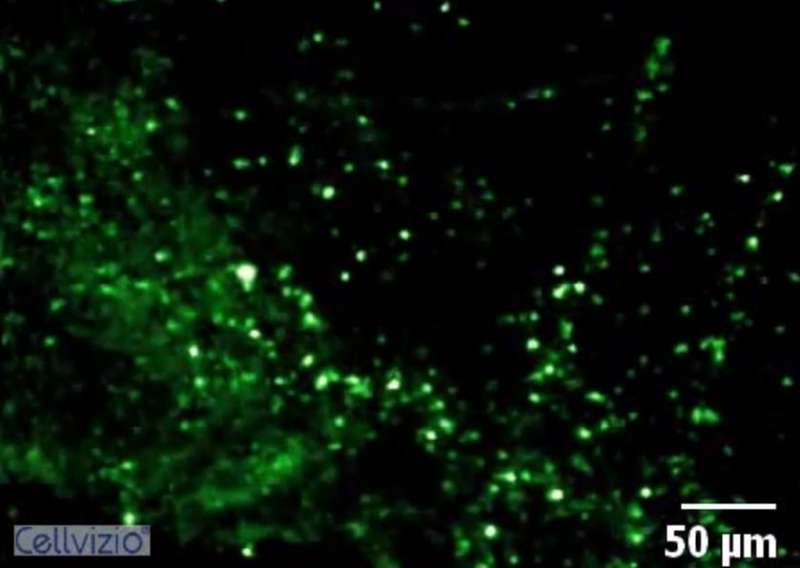
The new imaging technology involves spraying chemical probes into patients' lungs that light up when they attach to specific types of infectious bacteria.
This fluorescence is detected using tiny fibre-optic tubes that travel deep inside patients' lungs.
Researchers focused on one of the new probes, designed to detect a subset of bacteria called Gram-negative bacteria. These are fast becoming the hardest infections to treat with antibiotics and are a major cause of pneumonia.
The team developed the technology in the lab and then tested the probe in patients with a disease called bronchiectasis, which causes repeated cycles of inflammation and infection. They also tested intensive care patients who had suspected infection.
Striking fluorescence was seen in lungs of patients with bronchiectasis who had Gram-negative bacteria in the deep parts of their lungs.
Bacteria were also successfully visualised in intensive care patients using the new technology with striking results. In some of these patients, conventional methods had returned negative results. The researchers say this is not surprising as the individuals were being treated with several types of powerful antibiotics.
The underlying research was supported by Wellcome and the UK's Engineering and Physical Sciences Research Council (EPSRC) through support of the Proteus consortium, which includes the Universities of Edinburgh, Bath and Heriot-Watt.
Proteus is also funded by CARB-X, the world's largest public-private partnership devoted to antibacterial early development research.
The research is published in the journal Science Translational Medicine.
Kev Dhaliwal, Professor of Molecular Imaging & Healthcare Technology at the University of Edinburgh and a Consultant Respiratory Physician, said: "Drug-resistant bacteria are fast emerging as the greatest threat to humanity"
"We urgently need to develop and test new ways to diagnose infections in patients and also to improve our understanding of human disease. Our interdisciplinary Proteus team is developing next-generation technologies to improve patient care"
"The teamwork required to deliver this bench to bedside technology has taken many years and we are incredibly grateful to the patients and families who allowed us to test these exciting approaches and now the follow-on funding to further develop and test this technology in intensive care units around the UK."
More information: A.R. Akram el al., "In situ identification of Gram-negative bacteria in human lungs using a topical fluorescent peptide targeting lipid A," Science Translational Medicine (2018). stm.sciencemag.org/lookup/doi/ … scitranslmed.aal0033