Regulating the immune system's 'regulator'
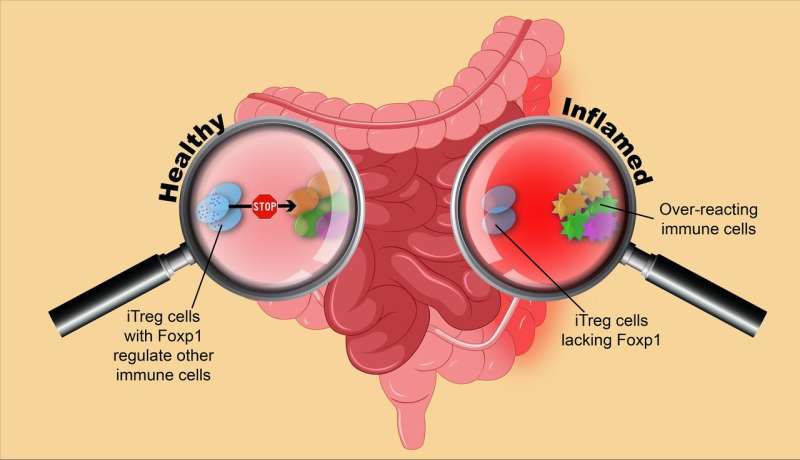
A research team at the Academy of Immunology and Microbiology, within the Institute for Basic Science (IBS) has discovered a possible therapeutic target that pulls the reins of immunity. In Nature Communications, the scientists reported that mice lacking Foxp1 protein in some specific immune cells are more susceptible to immune-induced inflammation.
The immune system is capable of protecting the body from invading pathogens, but it can also overreact and cause allergies and autoimmunity. In this respect, regulatory T (Treg) cells are an important built-in "self-check" of the immune system, preventing excessive immune reactions from happening. Since their discovery, Treg cells have been studied extensively as compelling and multifaceted therapeutic supplement. For example, Treg cells could block unruly immune responses in autoimmune diseases like Crohn's disease or inflammatory bowel disease. Vice versa, in the case of tumors, Treg cells would need to be restrained to let the anti-tumor immune response act more freely.
In the past decade, it has been largely established that two different types of Treg cells exist in mammals: thymic Treg (tTreg) and induced Treg (iTreg). tTreg cells are generated at a neonatal age in the thymus, and prevent fatal autoimmune reactions. By contrast, iTreg cells form later in life, and accumulate predominantly at mucosal barriers, such as the intestine, lungs, etc. In the large intestine, iTreg cells are essential to prevent an overenthusiastic immune response to fight against useful gut bacteria and harmless food.
iTreg cells could be good candidates for therapeutic interventions. iTreg precursors, unlike tTreg cells, could potentially be isolated from the host, modified, expanded in the laboratory, and subsequently transferred back to the host. However, researchers are facing problems with iTreg's stability: Once generated, these cells tend to lose their regulatory identity and convert to other cell types.
In this study, IBS researchers found that Foxp1 provides stability to iTreg cells by interacting with the gene encoding Foxp3 – the protein that confers identity to Treg cells. iTreg cells lacking Foxp1 gradually lose Foxp3, and become unable to perform their regulatory functions against excessive immunity. In mice, the lack of Foxp1 in iTreg cells led to intestinal inflammation and increased susceptibility to colitis.
Moreover, IBS biologists showed that Foxp1 is essential for sustaining optimal expression of Foxp3 specifically in iTreg cells, not in tTreg cells. "In a venture to spot differences hidden behind cells with similar appearances, we found Foxp1. The results suggest that this protein could be used to engineer more stable iTreg cells, while keeping their thymic counterparts untouched," explains Dipayan Rudra, corresponding author of the study.
More information: Sayantani Ghosh et al. The transcription factor Foxp1 preserves integrity of an active Foxp3 locus in extrathymic Treg cells, Nature Communications (2018). DOI: 10.1038/s41467-018-07018-y