Researchers discover key gene in cells associated with age-related hearing loss

An international group of researchers, led by Ronna Hertzano, MD, Ph.D., Associate Professor, Department of Otorhinolaryngology-Head & Neck Surgery, Anatomy and Neurobiology, at the University of Maryland School of Medicine (UMSOM), and Michael Bowl, Ph.D., Programme Leader Track Scientist, Mammalian Genetics Unit, MRC Harwell Institute, UK, have identified the gene that acts as a key regulator for special cells needed in hearing.
The discovery of this gene (Ikzf2) will help researchers better understand this unique type of cell that is needed for hearing and potentially develop treatments for common age-related hearing loss.
"Outer hair cells are the first inner ear cells lost as we age. Age-related hearing loss happens to everyone. Even a 30-year-old has lost some of the outer hair cells that sense higher pitch sounds. Simply exposure to sound, especially loud ones, eventually causes damage to these cells," said Dr. Hertzano, whose research will be published in the journal Nature.
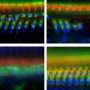
The inner ear has two kinds of sensory hair cells required for hearing. The inner hair cells convert sounds to neural signals that travel to the brain. This compares to outer hair cells, which function by amplifying and tuning sounds. Without outer hair cells, sound is severely muted and inner hair cells don't signal the brain. Loss of outer hair cells is the major cause of age-related loss of hearing.
About the Research
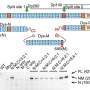
Hertzano's group, in collaboration with Ran Elkon, Ph.D., Senior Lecturer, Department of Human Molecular Genetics and Biochemistry, Sackler Faculty of Medicine in Tel Aviv, Israel took a bioinformatics and functional genomics approach to discover a gene critical for the regulation of genes involved in outer hair cell development. Bowl's group studied mice from the Harwell Aging Screen to identify mice with hearing loss. Bowl identified mice with an early-onset hearing loss caused by an outer hair cell deficit. When the two groups realized that they were studying the same gene, they began to collaborate to discover its biological function and role in outer hair cell development. The gene is Ikzf2 gene, which encodes helios. Helios is a transcription factor, a protein that controls the expression of other genes. The mutation in the mice changes one amino acid in a critical part of the protein, which impaired the transcriptional regulatory activity of helios in the mice.
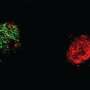
To test if helios could drive the differentiation of outer hair cells, the researchers introduced a virus engineered to overexpress helios into the inner ear hair cells of newborn mice. As a result, some of the mature inner hair cells became more like outer hair cells. In particular, the inner hair cells with an excess of helios started making the protein prestin and exhibited electromotility, a property limited to outer hair cells. Thus, helios can drive inner hair cells to adopt critical outer hair cell characteristics.
Funding for the research was provided by Action on Hearing Loss UK, a National Hearing Loss Charity, the National Institute on Deafness and Other Communication Disorders (NICD) at the National Institutes of Health, and the Department of Defense, USA.
As Professor Steve Brown, Ph.D., Director, MRC Harwell Institute, comments, "The development of therapies for age-related hearing loss represents one of the big challenges facing medicine and biomedical science. Understanding the genetic programs that are responsible for the development and maturation of sound-transducing hair cells within the inner ear will be critical to exploring avenues for the regeneration of these cells that are lost in abundance during age-related hearing loss. The teams from the University of Maryland and the MRC Harwell Research Institute have given us the first insights into that programme. They have identified a master regulator, Ikzf2/helios, that controls the programme for maturation of outer hair cells. Now, we have a target that we can potentially use to induce the production of outer hair cells within damaged inner ears, and we are one step closer to offering treatments for this disabling condition."
More information: Lauren Chessum et al, Helios is a key transcriptional regulator of outer hair cell maturation, Nature (2018). DOI: 10.1038/s41586-018-0728-4