Researchers report key to efficient production of heart muscle from pluripotent stem cells

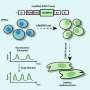
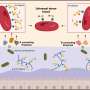
The transformation of embryonic stem cells, and their induced pluripotent counterparts into potentially limitless supplies of new heart tissue has been hindered by a lack of consistency from one stem cell line to the next, as well as a poor understanding of the differentiation process. Now, A*STAR scientists have shown that the key to efficient cardiac cell production lies in syncing the induction of differentiation with the cell cycle of the starting pluripotent material.
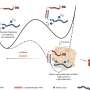
Although a widely used protocol for making heart muscle seems to work better with some stem cell lines than others, Steve Oh and his colleagues from the A*STAR Bioprocessing Technology Institute found this same standard method can be made to work with all stem cells with high efficiency if the cells are first cultured at optimal densities and stages of the cell cycle ahead of cardiac induction.
"A stem cell bioprocess scientist, once understanding what to control, can differentiate any pluripotent stem cell line efficiently," Oh says. "You must have actively cycling cells when you start differentiation—then efficiencies will increase."
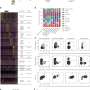
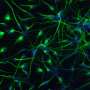
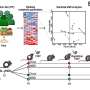
Discovering these lessons required Oh and his team to systematically try differentiating five pluripotent stem cell lines into heart muscle tissue. The researchers varied the culture conditions and administered a drug that directs cells down a cardiac lineage. They found that cells grown in a less crowded environment and in the DNA-replicating or active division stages of the cycle responded favorably to the drug molecule and adopted a cardiac profile. By comparison, those lines with dense clusters of cells in the earliest quiescent phase of the cycle tended to die off in response to the drug treatment.
Individual stem cell lines are not locked into particular rhythms. And by altering culture conditions or the timing and dose of the inducer drug, the researchers managed to devise protocols that worked reliably for each of the five lines.
The findings, says Oh, point to the need for scientists to fine-tune their differentiation methods based on the unique cell cycle profiles of their stem cell lines if they wish to obtain consistent, high-quality heart muscle cells in this way with an efficient yield. Their results could make it easier for researchers to study human heart development in a lab dish, discover new treatments for cardiac disease and, ultimately, develop new regenerative therapies for people with life-threatening heart problems.
More information: Filip Laco et al. Unraveling the Inconsistencies of Cardiac Differentiation Efficiency Induced by the GSK3β Inhibitor CHIR99021 in Human Pluripotent Stem Cells, Stem Cell Reports (2018). DOI: 10.1016/j.stemcr.2018.03.023