Scientists tackle rare retinal disease in unique research project
Researchers at the Lions Eye Institute (LEI) and The University of Western Australia have pioneered a study of the world's first and only laboratory model of a rare type of retinal disease, paving the way for new treatment pathways.
Cone dystrophy with supernormal rod response (CDSRR) is an inherited disease which emerges in childhood and will eventually cause life-long visual loss. Symptoms include central vision loss, extreme sensitivity to light, night and colour blindness and blind spots.
Around one in a million people are affected by CDSRR and there are currently no therapies available.
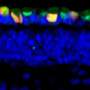
A team of researchers from the LEI and UWA, led by Emeritus Professor David Hunt and Dr. Livia Carvalho, have published new research in eNeuro which describes and validates a laboratory model of CDSRR.
"We believe this model closely matches what we see in humans with CDSRR and we will be able to use this to validate treatment strategies for CDSRR, better understand the physiology of vision and the pathophysiology of this disorder," Dr. Carvalho said.
The model was developed by the Welcome Trust Sanger Institute and brought to Western Australia from the United Kingdom by Professor Hunt in 2013.
While still in the United Kingdom, Professor Hunt led the first study that linked the genetic cause of CDSRR to mutations in a gene called KCNV2. The gene specifies a channel responsible for the movement of potassium ions across cell membranes in the retina.
"Since the publication of our original study in 2006, around 40 different disease-causing mutations have now been identified worldwide," Emeritus Professor Hunt said.
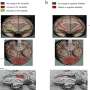
CDSRR is diagnosed using a test called an electroretinogram, which measures the electrical responses of various cell types in the retina. Affected individuals show very depressed rod and cone cell activity which switches to a supernormal rod response when presented with a very bright flash of light.
"So far, CDSRR is the only vision loss disorder that can be diagnosed without genetic screening because the supernormal electroretinogram response is unique to this disease," Emeritus Professor Hunt said.
"With this new study we have now confirmed that this is also the case in our laboratory model of CDSRR".
As the only group in the world working with this unique model, Emeritus Professor Hunt and Dr. Carvalho are in a prime position to develop and test treatment strategies.
"Due to the recessive inherited nature of CDSRR and the type of cells that are affected, we believe this disease would be an ideal candidate for a gene therapy based treatment," Dr. Carvalho said.
"We are currently working towards attracting funds to continue our work, not only to test treatments but also to understand the biology behind the supernormal rod response."
More information: Nathan S Hart et al. The role of the voltage-gated potassium channel proteins Kv8.2 and Kv2.1 in vision and retinal disease: insights from the study of mouse gene knock-out mutations, eNeuro (2019). DOI: 10.1523/ENEURO.0032-19.2019