Study merges big data and zebrafish biology to reveal mechanisms of human disease

In a series of studies that volleyed between large databases and research in zebrafish, Vanderbilt investigators have discovered a link between vascular biology and eye disease.
The research uncovered an unexpected role for the gene GRIK5, and it showcases a new paradigm for using biobanks, electronic health records and zebrafish to discover the genetic mechanisms that contribute to human disease. The findings were reported in the American Journal of Human Genetics.
The back-and-forth approach primarily involved two teams of investigators in the Division of Genetic Medicine: Eric Gamazon, Ph.D., and Nancy Cox, Ph.D., using computational genetics and large databases, and Gokhan Unlu, Ph.D., and Ela Knapik, MD, studying gene function in zebrafish.
"This iteration between statistical genetics analyses and biobanks and studies in animal models provides a framework that can be used to understand what a gene does within the context of the human phenome—all the diseases and traits included in electronic health records," said Gamazon, research instructor in Medicine.
The studies had their start several years ago when Gamazon and Cox, Mary Phillips Edmonds Gray Professor of Genetics and director of the Vanderbilt Genetics Institute, developed a computational method called PrediXcan that correlates genetically regulated gene expression with phenotypes. In the current work, they applied PrediXcan to BioVU, Vanderbilt's DNA biobank and de-identified electronic health records, to generate a comprehensive catalog of associations between gene expression and clinical traits.
Among the associations in this catalog, which they call PredixVU, the gene GRIK5 stood out. Reduced expression of GRIK5, which encodes a glutamate receptor subunit, was associated with 18 different eye diseases ranging from retinal detachment to cataract to glaucoma.
"This was very unusual—the expectation was that a gene might be associated with four or five disease phenotypes," said Knapik, associate professor of Medicine and Cell and Developmental Biology.
The relationship of GRIK5 to eye phenotypes was also unexpected, Knapik added. GRIK5 has mostly been studied for its role in brain signaling and cognition.
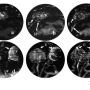
To explore the role of GRIK5 in zebrafish, Unlu used two approaches (CRISPR gene editing and morpholino oligonucleotides) to delete or reduce expression of GRIK5. The most striking finding in the zebrafish with reduced or no GRIK5 was bleeding.
"This was a complete surprise. There was no previous knowledge that GRIK5 had anything to do with the blood or vasculature," Knapik said.
Gamazon and Cox returned to the databases to search for evidence of patients who have both eye and vascular diseases.
They analyzed electronic health records for more than 2.6 million Vanderbilt subjects and found significant comorbidity of eye and vascular diseases, and they confirmed this comorbidity using an insurance claims dataset of 150 million individuals.Unlu and Knapik continued their studies in zebrafish. They used the morpholino approach to reduce expression of GRIK5 in a zebrafish line with glowing green blood vessels and found reduced numbers of blood vessels and other vascular abnormalities in the eye, brain and trunk. They labeled the plasma with nanobeads and found leaking blood vessels in the eye, brain and ear.
"Our studies show that not only the architecture of the vasculature but also the function—keeping the blood inside—is compromised in zebrafish with reduced GRIK5 expression," said Unlu, research fellow in Genetic Medicine.
In further computational studies of genotyped BioVU participants, Gamazon and Cox demonstrated an association of reduced genetically predicted GRIK5 expression with comorbid vascular and eye diseases.
The researchers propose that reduced GRIK5 expression compromises vascular perfusion to the eye, leading to late-onset diseases like cataracts and glaucoma.
Further studies are needed to understand the precise role of GRIK5 in the vasculature—and how defects lead to eye diseases, but the framework from these studies can be broadly applied, Knapik said.
"I am a developmental geneticist. We do forward genetics in zebrafish: we make changes in the genome and study the phenotypes that result," she said. "On the human side, Mother Nature has made changes in our genomes. And the health care system is phenotyping every day and keeping the results in electronic health records. Scientists can now use the approach we've demonstrated to understand the mechanisms of human disease."
More information: Gokhan Unlu et al, GRIK5 Genetically Regulated Expression Associated with Eye and Vascular Phenomes: Discovery through Iteration among Biobanks, Electronic Health Records, and Zebrafish, The American Journal of Human Genetics (2019). DOI: 10.1016/j.ajhg.2019.01.017