Mitochondrial permeability plays a key role in aging, recovery from ischemic injury
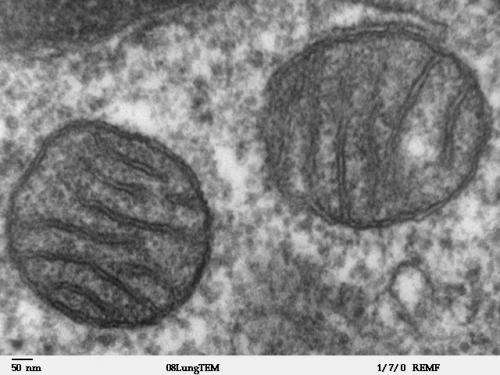
The ability of molecules to pass through the membrane of mitochondria—the cellular structures that convert nutrients into energy—may determine whether or not autophagy, a cellular process that removes damaged and dysfunctional molecules and cellular components, is beneficial or detrimental to the health of an organism. The results of a study led by Massachusetts General Hospital (MGH) investigators may improve understanding of aging-related dysfunction and could help prevent the tissue damage that results when the blood supply is cut off to vital organs like the heart and brain, as occurs in heart attacks and strokes.
"We have found for the first time that the 'leakiness' or permeability of mitochondria determines whether autophagy will extend or shorten lifespan," says Alexander Soukas, MD, Ph.D., of the MGH Center for Genomic Medicine, senior author of the report that has been published in Cell. "This raises that possibility that targeting mitochondrial permeability in aging and disease may maximize the health benefits of autophagy."
As the accumulation of damaged molecules and defective proteins is considered a hallmark of aging, autophagy has been associated with increased longevity. In fact, model organisms in which gene mutations or measures such as calorie restriction lead to lifespan extension depend on autophagy for their beneficial effects. However, autophagy can also play a role in cancer, diabetes, neurodegeneration and in the ischemia/reperfusion injury caused by restricted blood flow.
Previous studies have suggested that inhibition of the mTORC2 molecular pathway, which controls several critical metabolic functions, shortens lifespan. Organisms in which mutations in mTORC2 or in the gene encoding its downstream effector protein SGK-1 have reduced lifespan also show increased autophagy. The MGH-led team set out to investigate why elevated autophagy, which is beneficial in most instances, is detrimental in mTORC2/SGK-1 mutant animals and to identify the key cellular determinants of the impact of autophagy on health and lifespan.
Their experiments revealed that inhibition of autophagy can restore a normal lifespan in mTORC2/SGK1 mutant C. elegans roundworms. They also found that SGK-1 can regulate the opening of the mitochondrial permeability transition pore (mPTP), which allows very small molecules to pass through the mitochondrial membrane. Excessive opening of the mPTP, either by inhibition of the mTORC2/SGK-1 pathway or by direct genetic stimulation, transforms autophagy from a beneficial to a detrimental function, resulting in a shortened lifespan. Overall, the results indicate that the beneficial effects of autophagy depend on low levels of mitochondrial permeability.
Since autophagy is believed to contribute to ischemic injury, the investigators looked at its potential role in ischemia/reperfusion (I/R) injury—the exacerbation of tissue damage that occurs when blood flow is restored to tissue to which it had been restricted. They found that mice in which expression of the gene for SGK-1 was knocked out in the liver were more susceptible to I/R injury of the liver than were unmutated animals. While both current and previous research has indicated that elevated autophagy and mitochondrial permeability are harmful in the early phases of reperfusion injury, autophagy may help reduce the severity of tissue damage at later stages when damaged cellular components must be cleared from the cell.
"We think our work has major implications for preventing ischemic injury during heart attack and stroke, as well as during organ transplantation, which requires temporary interruption of the donor organ's blood supply" says Soukas, an assistant professor of Medicine at Harvard Medical School. "These processes all share features with the model used in our study, which implies that targeting mitochondrial permeability through mTORC2, SGK-1 or the mPTP has great potential for reducing the potentially devastating outcome of those occurrences."
Overall, he adds, the current study has provided the first direct evidence that elevated mitochondrial permeability reduces lifespan and blocks the beneficial effects of autophagy on health. They also provide direct genetic evidence that low levels of mitochondrial permeability are required to increase longevity and improve health and identify SGK-1 as an important regulatory molecule, modulation of which may help increase lifespan and reduce age-associated disease.
Soukas says, "In order to fully appreciate how mitochondrial permeability goes awry in aging, we need to understand the full spectrum of aging-related changes that increase mPTP opening and the mechanisms by which it contributes to age-related decline and disease. Understanding how health-promoting pathways successfully reduce mitochondrial permeability may provide new insights to promote healthy aging."
More information: Ben Zhou et al, Mitochondrial Permeability Uncouples Elevated Autophagy and Lifespan Extension, Cell (2019). DOI: 10.1016/j.cell.2019.02.013