Anaphylactic shock: Anti-drug IgG antibodies and neutrophils play an unexpected role
Anaphylactic shock, an exacerbated allergic reaction that can prove fatal, is sometimes caused by the use of drugs during surgery. In most of these extreme reactions, evidence can be provided that patients have anti-drug antibodies of the IgE class. In 10 to 20 percent of anaphylactic cases, evidence for the involvement of anti-drug IgE is lacking.
Anti-drug IgE enable activation of mast cells and basophils that release histamine, a potent mediator involved in anaphylaxis. Teams from the Institut Pasteur, Inserm, the Paris Public Hospital Network (AP-HP), the CNRS, Paris-Sud University and Sorbonne University have successfully identified a new pathological mechanism responsible for these previously unexplained cases involving neutrophils activated by antibodies of the IgG class. These findings, published on July 10 in the journal Science Translational Medicine, will help improve diagnosis and treatment for patients with this type of shock.
Anaphylaxis is a hyperacute allergic reaction caused by an inappropriate immune response following the introduction of a usually harmless antigen into the body. When this antigen binds to antibodies already in the body, it triggers the secretion of large quantities of potent vasodilating mediators, sending the patient into a state of shock that may result in multiple organ failure and even death.
Anaphylaxis may be brought about by various substances, including drugs (antibiotics or neuromuscular blocking agents), food and insect venom. In this study, the scientists focused on allergic reactions to neuromuscular blocking agents (NMBAs), drugs used during general anesthesia to induce muscle relaxation. The incidence of anaphylactic shock caused by NMBAs is one case in every 10,000 to 20,000 surgeries, representing approximately three to five cases each week in the Greater Paris area.
Although it was already known that IgE antibodies could cause anaphylaxis, teams from the Institut Pasteur, Inserm, the Paris Public Hospital Network (AP-HP), the CNRS, Paris-Sud University and Sorbonne University have demonstrated in a clinical study that IgG antibodies can also be involved in drug-induced anaphylactic shocks. This unexpected role of IgG antibodies had already been identified in mice in 2011 by some of the same authors.
This multicenter study, led by Bichat Hospital (part of the Paris Public Hospital Network), was launched in 2012 by a consortium of scientists, clinical biologists and anesthetists. The consortium monitored 86 patients with perioperative anaphylactic shock and 86 control patients in 11 hospitals in the Greater Paris area, coordinated at Bichat Hospital by immunologist Professor Sylvie Chollet-Martin (Paris-Sud University) and anesthetist Professor Dan Longrois.
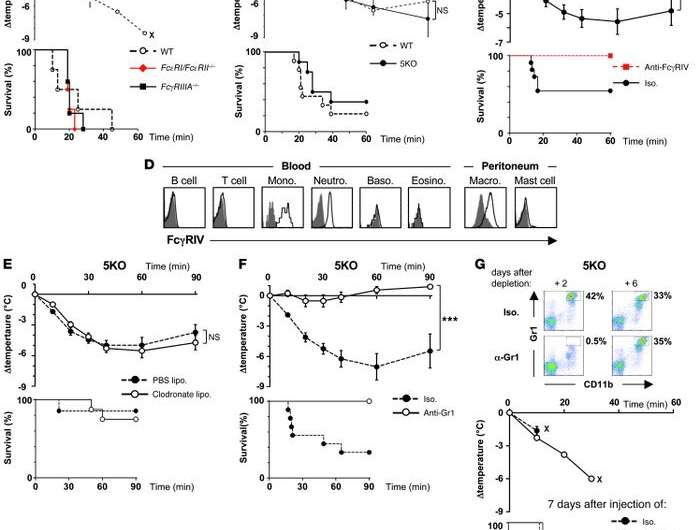
Blood samples were taken as soon as an anaphylactic shock occurred in the operating room, enabling the scientists to identify the alternative IgG-dependent mechanism. They demonstrated that IgG antibodies activate neutrophils (50-70 percent of our white blood cells), releasing high doses of harmful vasodilating mediators. Neutrophil activation was more pronounced in cases of severe shock than in cases of moderate shock. Interestingly, the IgG-neutrophil pathway was also identified in most cases of shock where the traditional IgE-dependent mechanism was observed, suggesting that IgGs and neutrophils may contribute to the severity of most cases of shock via an additive effect.
"These findings elucidate 10 to 20 percent of cases of anaphylactic shock that previously had no biological explanation. They will be extremely valuable in refining diagnosis in these patients and avoiding any future exposure with the drug that triggered the allergic reaction," explains Professor Sylvie Chollet-Martin (Paris-Sud University), joint last author of the study and Head of the Immunology laboratory on Autoimmunity and Hypersensitivity at Bichat Hospital.
"Although IgG antibodies are known to protect the body from infection and to act as aggressors in some autoimmune diseases, this study reveals that they may be involved in humans in another reaction that is harmful for the body, anaphylaxis. We are currently carrying out experimental research to explore how we might block this new activation pathway for IgG antibodies so that we can propose a therapeutic solution," comments Pierre Bruhns, joint last author of the study, Inserm Research Director and Head of the Institut Pasteur's Antibodies in Therapy and Pathology Unit.
More information: Friederike Jönsson et al. Mouse and human neutrophils induce anaphylaxis, Journal of Clinical Investigation (2011). DOI: 10.1172/JCI45232
F. Jönsson el al., "An IgG-induced neutrophil activation pathway contributes to human drug-induced anaphylaxis," Science Translational Medicine (2019). stm.sciencemag.org/lookup/doi/ … scitranslmed.aat1479