New target identified to combat deadly allergic reactions
Researchers in France have identified a molecular motor that controls the release of inflammatory factors that cause severe and fatal allergic reactions. The study, "Kinesin-1 controls mast cell degranulation and anaphylaxis through PI3K-dependent recruitment to the granular Slp3/Rab27b complex," which will be published online October 24 ahead of issue in The Journal of Cell Biology, suggests that targeting this motor may be a new way to treat patients undergoing anaphylactic shock.
Mast cells are a type of white blood cell localized in most tissues of the body that fight off parasitic infections by releasing histamine and other inflammatory factors contained in secretory granules. But allergens can also trigger granule release, and, if this happens simultaneously in mast cells throughout the body, it can provoke a life-threatening anaphylactic response that leads to facial swelling, difficulty breathing, a drop in blood pressure, and fainting. Currently, the main treatment for anaphylaxis is to immediately inject patients with epinephrine using devices such as the EpiPen from the pharmaceutical company Mylan NV.
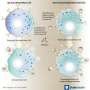
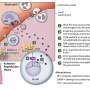
When mast cells are activated by allergens bound to immunoglobulin E antibodies, secretory granules move along microtubules to the periphery of the cells, where they can fuse with the plasma membrane and release their contents. Two teams of researchers led by Ulrich Blank (CNRS) of the Center of Research on Inflammation (CNRS) and Gaël Ménasché (INSERM) of the Imagine Institute in Paris, France, investigated the role of kinesin-1, a motor protein that transports various cargoes along microtubules.
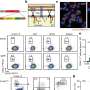
The researchers created mice whose mast cells lacked a key subunit of kinesin-1 called Kif5b. These animals were much less sensitive to allergen-induced anaphylaxis. Their mast cells showed lower levels of secretory granule release because the granules were no longer transported to the plasma membrane upon mast cell activation.
The researchers discovered that, in response to mast cell stimulation, a signaling pathway involving the enzyme phosphatidylinositol 3-kinase triggers kinesin-1's association with a protein complex on the surface of secretory granules. One member of this complex, Rab27b, has previously been shown to regulate mast cell secretion. Blank and Ménasché's team found that depleting the other member, Slp3, also impaired secretory granule transport and release, similar to the effects of removing Kif5b.
"The fact that mice lacking Kif5b in their mast cells exhibited very low levels of passive, systemic anaphylaxis suggests that kinesin-1 could be a valuable new therapeutic target for controlling allergic reactions," Ménasché says.
More information: Munoz, I., et al. 2016. J. Cell Biol. DOI: 10.1083/jcb.201605073