Expert panel in macular degeneration recommends paradigm shift for future directions
A panel of investigators assembled by the National Advisory Eye Council (NAEC) calls for large-scale collaborative research to address dry macular degeneration—the leading cause of blindness among the elderly—for which there is currently no effective treatment.
The NAEC—a 12-member panel that helps guide the National Eye Institute—recognized the need to investigate the current state of research on this disease, in order to guide the direction in which to take future research. The article, published in Nature Communications, is the product of a group of scientists tasked by the NAEC to do just that, as well as propose ideas on how to expand knowledge of this disease.
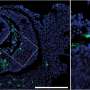
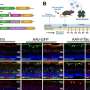
There are two types of age-related macular degeneration (AMD). "Wet" (neovascular) AMD, for which there are effective treatments that target limiting the inappropriate growth of new blood vessels that end up disrupting vision in the delicate tissues of the eye. "Dry" (non-neovascular) AMD is a more gradual vision loss from deposits of a substance called drusen, composed of fats and proteins. The proposed causes of dry AMD are numerous and are not all clear to the scientific and medical community.
"There are a plethora of genetic and environmental factors interweaving and contributing to this disease in ways known and unknown," explained co-corresponding author Lindsay Farrer, Ph.D., chief of the Biomedical Genetics division at Boston University School of Medicine. "Our article reflects on what we know thus far and calls for interdisciplinary research, with ideas on where to go next, towards answers that will bring about meaningful clinical results."
Among the ideas proposed in the article are drawing forth "integrated collaboration of leading clinicians, imaging experts, a wide variety of basic scientists, bioinformaticians and biostaticians" as well as creating a large biorepository of eye tissue from donors with and without AMD, generating multiple types of 'omics data from disease and normal eye tissue, and designing computer models of the disease. "Success in these areas will likely be achieved most expediently and effectively by promoting collaborative efforts of multidisciplinary investigator teams and developing centralized resources including clinical, imaging, omic, and other types of data as well as carefully phenotyped eye tissue from large cohorts of patients with and without AMD."
The authors also ask agencies such as the National Institutes of Health to reconsider how they allocate funding in order to make this possible. In addition, the authors suggest longer follow-up times with clinical trial patients, given there likely are different factors at play at different stages of the disease.