Single protein plays important dual shipping role in the brain
Just as a packaging breakdown can hamstring delivery of cables, switches and connectors to a house under construction, removing a protein from neurons can block the "shipment" of proteins to developing axons.
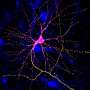
Axons are the telephone wires of the nervous system. They convey information to dendrites on other nerve cells, in a processing network of phenomenal complexity that is the backbone of the entire nervous system.
In a paper published Aug. 6 in Nature Communications, Edwin Chapman of the Howard Hughes Medical Institute and the University of Wisconsin–Madison reports that halting production of synaptotagmin 17 (syt-17) blocks growth of axons.
Equally significant, when cells made more syt-17, axon growth accelerated. A wide range of neurological conditions could benefit from the growth of axons, including spinal cord injuries and some neurodegenerative diseases.
The protein in question, syt-17, is made by the 17th (and last) synaptotagmin gene to be identified.
"Lots of work has been done on this family since it was discovered in 1981," Chapman says.
In many cases, synaptotagmin proteins serve as calcium sensors that trigger the release of chemical messengers called neurotransmitters, which nerve cells use to communicate, when calcium ions are present.
"Calcium ions are a basic signal in the nervous system, and so synaptotagmin proteins have been intensely studied," Chapman says.
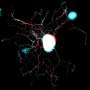
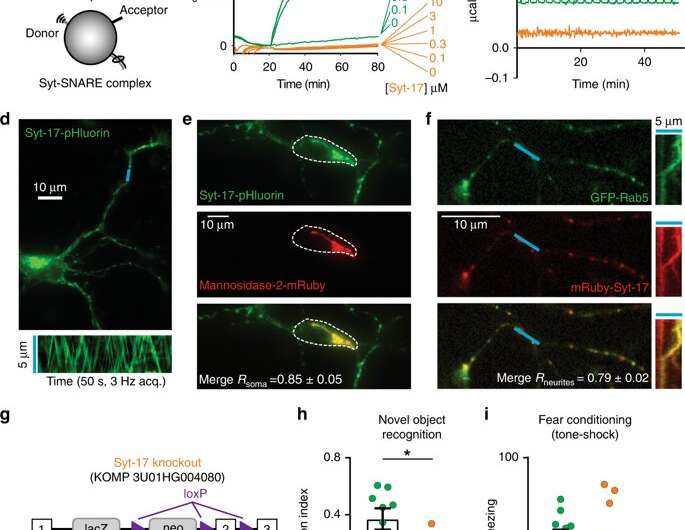
In a search to locate synaptotagmin proteins in neurons, Chapman and first author David Ruhl, then his graduate student, traced syt-17 to the Golgi apparatus. The Golgi is a shipping center inside the neuron that "packages" proteins for delivery from another part of the cell to the end of an axon, where growth occurs.
"It's a bit of a simplification," says Chapman, a professor of neuroscience, "but basically, you can't build without supplies, and one of the ways that neurons are able to build such long, complicated axons is through syt-17 speeding up the production line."
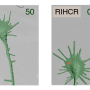
The key observation relating axon growth to syt-17 occurred about six years ago, when Chapman's lab was doing basic work to find the different synaptotagmin proteins inside neurons. "We made an accidental discovery that it makes axons grow really long," Chapman says. "Well, that was interesting! We decided to work on it."
One standard way to learn what a gene does is to "knock out," or silence, it. In syt-17 knockout mice, the axons barely grew, Chapman says. "But in mice genetically programmed to make an abnormally large quantity of syt-17, the axons grew much faster than normal."
The interaction is much like a construction project, Chapman says. "To grow an axon, you've got to send a lot of stuff down pipelines that supply the growing end of an axon. Think of building a house: You need shipments of studs, floor joists and roof shingles. A growing axon needs its own parcels, though they are much smaller."
In 2016, Ruhl discovered a second pool of syt-17 in the neuron. Finding two stashes "was weird," Chapman says. Ruhl began to notice that the protein had a split personality and eventually discovered it does two completely unrelated things in the same cell.
While the first stash was on the signal-shipping side, the second pool of syt-17 was in the dendrite, the signal-sensing side of the synapse. The synapse is the communication junction between two neurons.
"It's the exact opposite of what we'd have guessed," says Ruhl, now a postdoctoral researcher at the University of California, San Diego. "I think the second function is pretty cool."
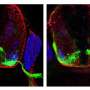
Ruhl eventually discovered that syt-17 at the dendrite tunes down synaptic communication by keeping a reserve of receptors inside the cell. Receptors bind to neurotransmitters in the synapse.
"Without syt-17 (at the dendrite) most of the receptors wind up on the surface and synapses are turned up to 11," he says.
This is not a bad thing in terms of brain plasticity—the ability of an adult brain to adapt and learn. "In plasticity, an important feature is increasing or decreasing receptivity to neurotransmitters," Chapman says.
Without this type of dampening, neurons could begin to fire uncontrollably, leading to problems like seizures.
And so syt-17 turns out to be "a key player in the negative half of the balance sheet," Chapman says. "It doesn't just help axons grow; it regulates how existing synapses respond to signals.
There's a certain amount of truth to the old saw that the brain, like a hard disk, needs to forget in order to make "room" for the new, Chapman says. "Remembering is important, but forgetting is important, too."
More information: David A. Ruhl et al. Synaptotagmin 17 controls neurite outgrowth and synaptic physiology via distinct cellular pathways, Nature Communications (2019). DOI: 10.1038/s41467-019-11459-4