UCSD's cancer vaccine trial shows range of results at the one-year mark
In April, one of the cancerous tumors in Carlos Gil's spine grew so large that it cracked his C-7 vertebra, causing so much pain that the father of four was forced to sleep in his downstairs guest bedroom, biting a pillow so that his kids wouldn't hear him screaming through the night.
In those long hours of agony, Gil said, it seemed like the end. But today, just four months after receiving a custom cancer vaccine engineered to target the specific genetic fingerprint inside his cancer cells, the Chula Vista resident said he is feeling better. Much better.
"Before the vaccine, I had a death sentence," Gil said. "I am proof that this can be successful."
Doctors at UC San Diego Moores Cancer Center say Gil's tumors are 90% smaller than they were when they were causing pain so severe that he sometimes passed out and paralysis seemed like a certainty.
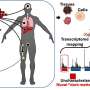
It's a miraculous turnaround and one that shows what's possible in the growing world of cancer vaccine research. Moores, working with immunologists from the La Jolla Institute of Immunology, is among a growing number of research institutions exploring this new oncological approach that uses genetic sequencing to identify unique mutations inside each patient's tumors, manufacturing special molecules called peptides that can program T cells, some of the immune system's most effective fighters.
For several years such trials have been showing intriguing hints that it is possible to precisely target malignant cells that other therapies like chemotherapy and radiation are unable to eradicate.
While most cancer vaccine trials to date have relied on sophisticated computer analysis to predict which mutations should be included in a vaccine, the approach that the Moores team is pursuing adds a functional step, testing predicted targets first, before they're included in a vaccine, to see if they have already been recognized by each patient's immune system. Such vetting, the team hopes, might elicit a stronger response.
So far, the results have been mixed.
Gil's response is by far the best among the five patients treated with the custom cancer vaccines over the past year.
Dr. Ezra Cohen, director of translational science at Moores, said that one patient died after her cancer shrugged off the vaccine, another withdrew from the trial and went back to chemotherapy, though the vaccine did seem to stop the tumors from growing. Another patient was just recently treated, and early results are not yet available.
Tamara Strauss, the trial's first participant—whose parents, Matthew and Iris Strauss, helped fund the trial with a $1 million donation—initially saw the tumors in her liver remain stable, but they didn't regress as they did with Gil. A biopsy showed that her tumors stopped expressing the two mutations that were targeted by her custom vaccine.
There were, Cohen noted, some tantalizing results, even though Strauss's tumors remain. While they didn't disappear, they also didn't grow.
"The fact that she had stable disease for about 10 months, I think that's evidence the vaccine was doing something, it just wasn't doing ideally what we wanted it to do," Cohen said.
A big part of the reason for that result, researchers suspect, is that the best target in Strauss's tumors—the one that looked like it would have the best chance of binding with molecules necessary for the target to be "presented" to unprogrammed T cells—wasn't included in her vaccine because the short molecules needed to incorporate the target didn't hold together during the manufacturing process.
But Cohen said that the project recently began working with a new vaccine manufacturer and took another stab at making the molecules that look like the best targets inside those tumors.
"We were excited to see that it worked the second time around, and so we're going to vaccinate her again, but we'll use a little different approach this time," Cohen said.
The initial study protocol called for three vaccine doses spaced three weeks apart, but Dr. Stephen Schoenberger, an immunologist at the La Jolla Institute of Immunology, said recent preclinical research has encouraged investigators to adopt a new dosing schedule going forward. The new approach will start with three vaccine doses given once per week for the first three weeks followed by between six and nine additional doses depending on a participant's immune response.
Schoenberger said the new approach is designed to mimic the way that infectious pathogens gradually stimulate the immune system, eventually eliciting a strong response. A new arm of the trial will be created to make a distinct record of patient responses under the new protocol so that results can be compared with the previous three-dose approach.
This is the normal experience of scientific investigation. Initial plans look good on paper, but need tweaking as results start coming back from real patients fighting the biggest battles of their lives.
Having already been through multiple rounds of chemotherapy and multiple abdominal surgeries, Strauss said she would, of course, have preferred that her first cancer vaccine caused her tumors to melt away. But, she said, she has no desire to pull back at this point.
"I really do feel like the next one is going to be the one," she said. "I think it's an honor to be a part of a process like this. I would have preferred not to have cancer at all, but, if I had to have it, at least I feel like I'm part of the process of trying to find a better way for all cancer patients."
The cancer struggle, she said, is simply barbaric for so many, and it's time to find better answers.
"This experience, it just crystallizes for me why I'm doing it," Strauss said. "This is no way for anybody to live. There has got to be a better way."
And that perspective is shared by the family of Kristin Peabody who died on Feb. 7 after her vaccine failed to help her immune system stop her parotid gland cancer. Kristin, said her mother Jan Heaton, started fund-raising efforts for the trial, establishing the UCSD Peabody Fund with her husband, Wyatt Peabody.
"We will continue to support Dr. Ezra Cohen and the personalized cancer vaccine initiative at UCSD," Heaton said in an email. "We believe this is the future of cancer treatment, as did Kristin. It is her legacy."
It's a rather nuanced vote of confidence for a family that did not see the result they were praying for. Cohen said such support, the ability to see the big picture amid the immediate pain of losing a loved one, that ultimately pushes science forward.
"I'm hard-pressed to think of any efforts in cancer research that were successful right out of the gate," he said. "Even the best therapies we have weren't universally successful the first time we tried them. The key, I think, is that we do things methodically, scientifically, and we learn as we go along."
It also helps to see what's possible. In Gil's case, it came down to five specially chosen targets called "neoantigens" by immunologists.
Though Gil is not yet back to work, the 43-year-old, who came to the United States 23 years ago, said he hopes to do so soon. His wife, Maria, was his rock on those painful sleepless nights, before the vaccine's furious action removed the relentless pressure from his over-stressed nerves, he said.
"No matter how bleak things looked, she always told me that I wouldn't die. She has always carried on as if they diagnosed me with a cold," Gil said.
Initially, when the Strauss family donated $1 million, the plan was to treat 10 patients, but only half of that number has received vaccines one year after Strauss received the first dose. Cohen said that's because it took a long time—two to three months—for a contract manufacturer to create the vaccines. The study has since found a new manufacturer capable of producing doses more quickly, which should allow the trial to reach 10 patients relatively quickly.
There are also plans to expand the trial to 20 patients. In addition to the Strauss and Peabody gifts, a $300,000 grant from Padres Pedal the Cause, a charity that has raised more than $10 million for cancer research, also helped cover increased costs. Cohen said current reserves will pay for a total of 15 patients, but the university will need to raise additional funds to cover the last five patients.
©2019 The San Diego Union-Tribune
Distributed by Tribune Content Agency, LLC.