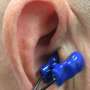
Electrical pulse to the ear reduces risk of post-operative atrial fibrillation
Atrial fibrillation is one of the most common complications following heart surgery, affecting one in three patients. This adverse side-effect, with symptoms such as palpitations, dizziness, shortness of breath or tightness in the chest, can normally be suppressed with sometimes strong medication. A Phase II study conducted at the Department of Surgery under the direction of Martin Andreas and Alfred Kocher, as well as Michael Wolzt at the Department of Clinical Pharmacology, has now shown that the risk of this complication could be halved by non-invasive electrical stimulation of the vagus nerve directly on the ear.
In addition to the usual risk factors such as advanced age, a history of atrial fibrillation or hypertension, other factors come into play following surgery, such as inflammatory reactions, oxidative stress and activation of the sympathetic nervous system, whose job it is to put the body into a heightened state of alert.
In its turn, this heightened state of alert increases the risk of atrial fibrillation following a heart operation. "Electrical stimulation of the ear and the vagus nerve that sits on the surface there has a calming effect, in that we are stimulating the largest nerve of the parasympathetic nervous system and hence the corresponding antagonists," explains lead author Martin Andreas from the Department of Surgery. This stimulation is given continuously in the first five days after an operation, when the risk of atrial fibrillation is at its highest, and is then stopped, if everything goes well.
This type of stimulation on the outer ear is non-invasive (the electrical pulse generator can be attached to clothing and transmitted via a cable to the ear, where the two electrodes are attached) and has never before been used for atrial fibrillation. The innovative product from an Austrian company from Mauerbach has already been successfully used for vascular occlusions.
Forty patients who had undergone heart surgery were included in this study at MedUni Vienna. "This is now being followed up by a Phase III study, in which we want to evaluate our promising results," says Martin Andreas. "Should this treatment prove to be successful in this indication as well, we can expect this innovation to be ready for clinical use in four or five years."
More information: Martin Andreas et al. Electrical Stimulation of the Greater Auricular Nerve to Reduce Postoperative Atrial Fibrillation, Circulation: Arrhythmia and Electrophysiology (2019). DOI: 10.1161/CIRCEP.119.007711