Influenza human challenge study begins at NIH-sponsored clinical trial units

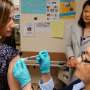
A clinical trial in which healthy adults will be deliberately infected with influenza virus under carefully controlled conditions is recruiting volunteers at four Vaccine and Treatment Evaluation Units (VTEUs) supported by the National Institute of Allergy and Infectious Diseases (NIAID). One study aim is to assess how levels of pre-existing influenza antibodies impact the timing, magnitude and duration of a volunteer's flu symptoms following exposure to influenza virus. NIAID is part of the National Institutes of Health.
The first doses of challenge virus were administered to five volunteers earlier this week. Up to 80 people aged 18 to 50 years will be enrolled in the trial. The study builds on recent work by scientists in the NIAID Laboratory of Infectious Diseases to develop a model of influenza disease with controlled human infection studies.
"NIAID investigators have been pioneers in contemporary human influenza challenge trials," said NIAID Director Anthony S. Fauci, M.D. "These trials provide a powerful tool to study many aspects of influenza disease progression and also can help to efficiently assess new treatments and vaccine candidates."
He added: "Expanding the capacity to perform human challenge trials is a key goal in NIAID's strategic plan to support the development of vaccines that confer broad and durable protection against influenza. This new trial at the VTEUs will help us achieve that goal."
Volunteers will receive a nasal spray containing a strain of seasonal influenza virus made under good manufacturing practice conditions. The challenge virus, InfluenzaA/Bethesda/MM2/H1N1, was developed by NIAID scientists and reliably produces mild to moderate influenza disease in most recipients. It has been administered to approximately 400 participants in four previous influenza challenge trials conducted at the NIH Clinical Center in Bethesda, Maryland. No significant safety issues or severe or complicated cases of influenza occurred, and no transmission of the virus outside of the clinic was seen during the earlier trials.
In the current trial, volunteers will remain in the clinic for at least seven full days after being challenged with the virus. Blood samples and nasal and throat swabs taken before and periodically after viral challenge will be used to trace the initiation, size and duration of various immune system responses, and to detect virus shedding. The appearance and resolution of flu symptoms, such as fever and muscle aches and weakness, will be recorded daily by the volunteers and study staff for a total of 14 days after the virus challenge. All volunteers will be followed for approximately 90 days post-challenge and will have additional blood and nasal wash samples taken at several follow-up clinic visits.
Preliminary trial results are expected in May 2020.
More information: Matthew J. Memoli et al. Validation of the Wild-type Influenza A Human Challenge Model H1N1pdMIST: An A(H1N1)pdm09 Dose-Finding Investigational New Drug Study, Clinical Infectious Diseases (2014). DOI: 10.1093/cid/ciu924