Newly engineered peptide shows potential as long-acting anti-HIV drug
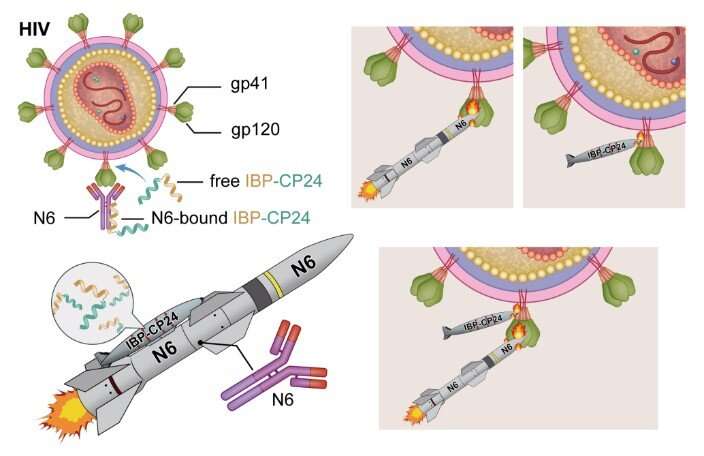
A newly engineered peptide called IBP-CP24 has the potential to be further developed as a long-acting anti-HIV drug that can be used alone or in combination with a broad neutralizing antibody for the treatment and prevention of HIV-1 infection, according to a study published December 5 in the open-access journal PLOS Pathogens by Lu Lu and Shibo Jiang of Fudan University and Lishan Su of the University of North Carolina at Chapel Hill, and colleagues. As reported in the study, IBP-CP24 exhibited a prolonged half-life as well as potent and broad anti-HIV-1 activity, even against drug-resistant strains.
Enfuvirtide is the first anti-HIV peptide drug approved by the U.S. Food and Drug Administration. However, its clinical application is limited because of its short half-life and the emergence of enfuvirtide-resistant HIV strains. In the new study, researchers developed a novel strategy to prolong the half-life of a short anti-HIV peptide called CP24 by fusing it to the human Immunoglobulin G (IgG) Fc-binding peptide (IBP).
IBP-CP24 inhibited a broad spectrum of HIV-1 strains, including those resistant to enfuvirtide. Most importantly, its half-life in the blood of rhesus monkeys was 46.1 h, approximately 26- and 14-fold longer than that of CP24 and enfuvirtide, respectively. IBP-CP24 intravenously administered in rhesus monkeys did not induce significant IBP-CP24-specific antibody response and showed no obvious toxicity. Mice pretreated with IBP-CP24 were protected from HIV-1 infection, and co-administration of IBP-CP24 and normal human IgG in mice with chronic HIV-1 infection resulted in a significant decrease in viruses in the bloodstream. Interestingly, the combined use of IBP-CP24 and a broad HIV neutralizing antibody showed a synergistic anti-HIV-1 effect, suggesting that this strategy may reduce the dose of the antibody and peptide and the cost of treatment.
More information: Bi W, Xu W, Cheng L, Xue J, Wang Q, Yu F, et al. (2019) IgG Fc-binding motif-conjugated HIV-1 fusion inhibitor exhibits improved potency and in vivo half-life: Potential application in combination with broad neutralizing antibodies. PLoS Pathog 15(12): e1008082. doi.org/10.1371/journal.ppat.1008082