New protein function could be key to treatment of drug addiction and behavioral disorders
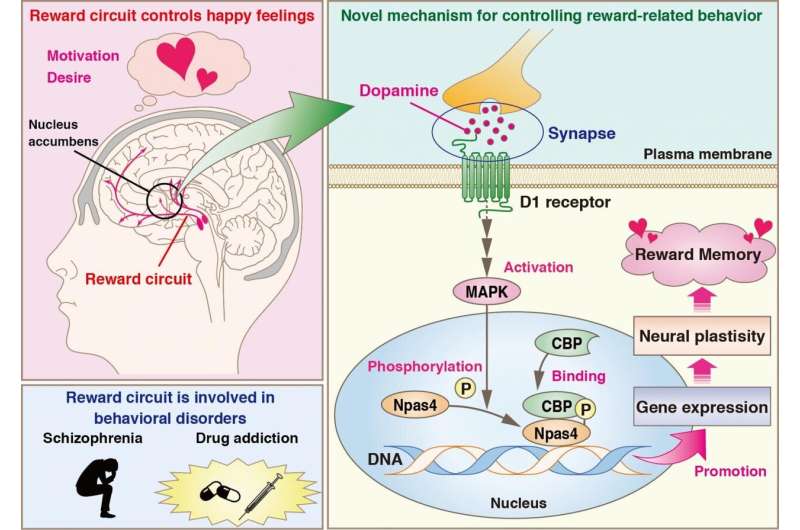
The human brain consists of several intricate networks or "circuits." One such complex circuit, called the "reward circuit," is involved in reward-associated learning, a process in which nerve cell activity changes in response to a "reward" stimulus (something that the brain perceives as a reward). This process is what usually causes feelings of desire and motivation, but excessive stimulus may also cause dependence and addiction.
The main regulator involved in the reward circuit is dopamine, a chemical popularly called the "happy hormone." In a region of the brain called nucleus accumbens, this chemical targets a specific group of neurons, called the medium spiny neurons, and induces a series of molecular changes. Unfortunately, irregularities in this pathway are associated with several behavioral and cognitive disorders, including Parkinson's disease, compulsive behavior, autism, and schizophrenia. Drug addiction, especially to potent stimulants like cocaine and amphetamines, is also dependent on this pathway. Unequivocally, to develop effective therapies, it is important to understand exactly how the reward pathway works.
In a new study published in Cell Reports, a team of scientists in Japan, led by Prof Kozo Kaibuchi and Dr. Yasuhiro Funahashi from Nagoya University, has identified a novel protein function involved in the reward circuit of the brain. They based their study on the fact that dopamine, when released in the brain, activates several proteins in response, and these proteins then cause certain changes in brain activity, such as reward-related gene expression and changes in nerve transmission or plasticity. But, how these changes happen at the molecular level was not well understood. The scientists at Nagoya University wanted to dig a little deeper. Prof Kaibuchi says, "Very little is currently known about how dopamine works in neurons to establish reward memory and cause addiction. This motivated us to pursue this study."
What was known so far was that dopamine activates multifunctional proteins, such as CREB-binding protein (CBP), which in turn promotes gene expression through interactions with other proteins. To find more detailed information, the scientists searched for proteins interacting with CBP in mice that experienced a conditioned reward. Using protein interaction-based experiments and database analyses, they successfully identified many such proteins. Of these, one protein, called Npas4, a "transcription factor" (a protein that binds to specific DNA sequences and regulates transcription from DNA to mRNA), was known to function in reward-related learning, and so the scientists moved on to find out its mechanism.
In a first for the scientific community, the group showed that a protein kinase called MAPK adds a phosphate group to Npas4 (a well-known intracellular process called phosphorylation, which "activates" proteins to carry out their functions), thereby facilitating its interaction with CBP. The scientists even identified the exact sites where MAPK phosphorylates Npas4. Moreover, they found that dopamine activates Npas4 in striatal medium spiny neurons, and so promotes the expression of genes related to "neural plasticity" (changes in neural connections). Thus, the three dopamine-stimulated proteins CBP, MAPK, and Npas4 interact with each other, leading to profound neural changes—Dr. Funahashi and Prof Kaibuchi had unraveled a major mechanism of how dopamine affects the brain.
To investigate the function of Npas4 in reward-related behavior, the scientists then "knocked out" or inactivated Npas4 in the reward circuit neurons of mice. These mice and normal mice learned to expect a cocaine reward in only one of two chambers, after which chamber preference was measured. Compared to normal mice, the Npas4-deficient mice showed a >50% reduction in drug-seeking behavior, indicating a significant reduction in reward memory. Importantly, the drug-seeking behavior was restored after exogenous administration of Npas4 but not by phospho-deficient mutants of Npas4. This was exciting, as it confirmed that Npas4 and its phosphorylation play an important role in reward-related behavior.
These unprecedented findings by Prof Kaibuchi and his team shed some light on the pathways involved in the reward circuit of the brain. Faulty functioning of the reward circuit is seen in various neuropsychological and cognitive disorders. By explaining in detail, the function and reward-associated mechanism of Npas4, these scientists have paved the way for new, effective therapies. Talking about the applications of the study, Prof Kaibuchi says, "Our study can help develop treatments for neuropsychiatric disorders such as schizophrenia. It can also be useful in tackling addiction to or dependence on cocaine and other stimulants."
Could a reward for this exciting study come in the form of a potential new treatment for behavioral disorders? Only time will tell. But, Prof Kaibuchi's team and their study certainly offer hope for the future.
More information: Yasuhiro Funahashi et al, Phosphorylation of Npas4 by MAPK Regulates Reward-Related Gene Expression and Behaviors, Cell Reports (2019). DOI: 10.1016/j.celrep.2019.10.116