Replacing animal testing with synthetic cell scaffolds
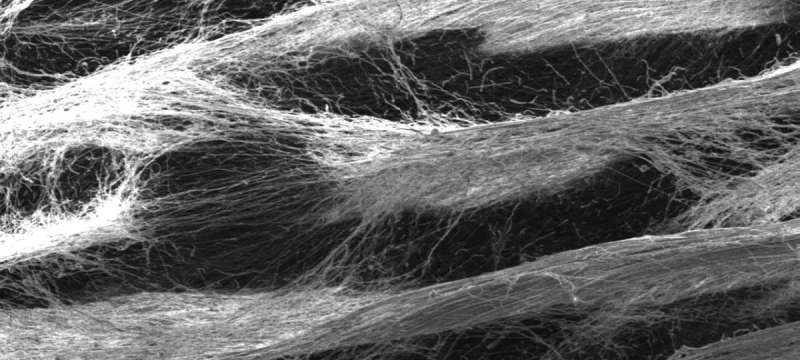
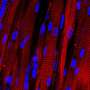
Electrospun synthetic cell scaffolds are not only more consistent than animal cells for cancer research, they hold the potential to replace animal testing.
In the field of cancer research, the idea that scientists can disrupt cancer growth by changing the environment in which cancerous cells divide is growing in popularity. The primary way researchers have tested this theory is to conduct experiments using animals.
Smitha Rao's cell scaffolding research aims to replace animal testing in cancer research with electrospun synthetics.
Rao, assistant professor of biomedical engineering at Michigan Technological University, recently published "Engineered three-dimensional scaffolds modulating fate of breast cancer cells using stiffness and morphology related cell adhesion" in the journal IEEE Open Journal of Engineering in Medicine and Biology.
Rao's coauthors are doctoral student Samerender Hanumantharao, master's student Carolynn Que and undergraduate student Brennan Vogl, all Michigan Tech biomedical engineering students.
Standardizing with Synthetics
When cells grow inside the body, they require something known as an extra-cellular matrix (ECM) on which to grow, just like a well-built house requires a strong foundation. To study how cells grow on ECMs, researchers need to source the matrices from somewhere.
"Synthetic ECMs are created by electrospinning matrices from polymers such as polycaprolactone and are more consistent for research than using cells from different kinds of animals," Hanumantharao said.
"In my lab the focus has been on standardizing the process and using synthetic materials to keep the same chemical formulation of a scaffold, but change the physical structure of the fibers that are produced," Rao said, noting that changing the type of polymer or adding solvents to polymers introduces too many variables, which could affect the way cells grow on the scaffolds. Rao and her fellow researchers, therefore, can compare separate cell lines with different scaffold alignments by changing just one aspect of the experiment: voltage.
By changing the voltage at which the polymer is spun, the researchers can alter the shape of the scaffolds, whether honeycombed, mesh or aligned. Rao's team published recently in Royal Chemistry Society Advances about manipulating electric fields to achieve different scaffold patterns. Rao's team is working with Dipole Materials to explore scaling up the process.
Rao and her fellow researchers used four different cell lines to test the efficacy of the electrospun scaffolds: 184B5, which is normal breast tissue, as a control; MCF-7, a breast adenocarcinoma; MCF10AneoT, a premalignant cell line; and MDA-MB-231, a triple negative adenocarcinoma-metastatic, a very difficult-to-detect cancer.
"We can study why and how cancer cells metastasize," Rao said. "We can understand in a true 3-D system why pre-metastatic cells become metastatic, and provide tools to other researchers to study signaling pathways that change between pre-malignant and malignant cells."
Avenues for Future Research
In addition, the research has uncovered information for another area of study: In what type of cellular environment do malignant cancer cells grow best? Rao's group discovered that the triple-negative breast cancer cells preferred honeycomb scaffolds while adenocarcinoma cells favored mesh scaffolds and premalignant cells preferred the aligned scaffolds. In the future, scientists may be able to engineer cell scaffolding—stiffness, structure and shape—to make the area around a tumor in a person's body a far less hospitable place for cancer cells to grow.
More information: Samerender Nagam Hanumantharao et al. Engineered three-dimensional scaffolds modulating fate of breast cancer cells using stiffness and morphology related cell adhesion, IEEE Open Journal of Engineering in Medicine and Biology (2020). DOI: 10.1109/OJEMB.2020.2965084