Neurodevelopment team locates a gene that helps control puberty and human reproduction

Thousands of Americans suffer from the delay or non-appearance of puberty. While the underlying cause is failure in the correct action of the hypothalamic hormone GnRH, the precise genetic causes have not been fully understood.
A UAlbany biologist and his research team have now identified a particular gene and its mutation as a possible culprit in adversely affecting puberty—the process of hormonal and physical changes that leads our body to reach sexual maturity and reproductive competence.
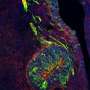
In a study published this month in the Journal of Neuroscience, Paolo E. Forni, assistant professor of Biology, and his team identified a key role for a transcription, called Gli3, in controlling the migratory process of the GnRH-1 neurons to the brain. Moreover, Forni's team, in collaboration with Ravikumar Balasubramanian of Massachusetts General Hospital and Gabriele Fuchs, assistant professor of Biology, was able to identify and functionally validate a new genetic mutation of the GLI3 gene in humans that is associated with a lack of puberty.
"The hormonal changes that initiate and control puberty are coordinated by Gonadotropic Releasing Hormone, or GnRH, a specialized population of neurons," said Forni. "The GnRH neurons modulate the release of signaling molecules from the brain that control the synthesis and release of sex hormones from the gonads—ovaries in females and testicles in males."
Forni points out that, during embryonic development, the GnRH are generated by stem cells in the nose and then migrate into the brain. Defective migration of the GnRH neurons to the brain negatively affects the production of sex hormones, the onset of puberty and therefore fertility.
"The data in our study have important clinical relevance as they provide new insights into GnRH1 development and suggest that human GLI3 mutations can contribute to reproductive disorders in humans," said Forni.
He notes that his team's work can lead to greater understanding of human reproductive syndromes and future remedies.
"Identifying the contribution of mutations of Gli3 in reproductive syndromes is an important step, first of all, for understanding the genetic causes of these syndromes, such as Kallmann Syndrome," he said. "Moreover, our results can improve diagnostic criteria and stimulate the development of novel treatments and therapeutic strategies to improve the human condition."