Battle with the cancer: New avenues from childhood vaccines
A new study from the University of Helsinki shows for the first time how the pre-immunization acquired through common childhood vaccines can be used to enhance therapeutic cancer treatment.
Professor Vincenzo Cerullo's research team at the University of Helsinki discovered that when animals were pre-immunized with ordinary vaccine, e.g. anti-tetanus, engrafted with tumor and then treated with the new hybrid viral platform called PeptiCRAd, they showed a dramatic improvement in tumor-specific immune response and, consequently, a significantly improved anti-tumor efficacy. According to Cerullo this approach can be very easily translated into clinical trials as it relies on pre-existing immunity of vaccines included in the national vaccination programs worldwide.
"This method has potential to have a significant impact on current immunotherapy protocols," says Vincenzo Cerullo, professor at University of Helsinki, director of the Drug Research Program and a founder at VALO therapeutics.
Due to the high coverage of international vaccination programs, majority of the worldwide population has been vaccinated against common pathogens, leading to a pathogen specific immunological memory. This is able to deploy a much faster and more effective immune response when we re-encounter the pathogens; this is called secondary response and it is stronger and faster than the first time we encounter a pathogen (primary response). Generally speaking, the therapeutic cancer vaccines generate an anti-tumor response more similar to a primary than a secondary immune response.
"To overcome this limitation, and convert the anti-tumor response into a secondary response we developed a hybrid tumor-pathogen therapeutic cancer vaccine," explains Cerullo. "Since their introduction, the vaccines have made one of the greatest contribution to public health, with the eradication of common deadly infections such as smallpox and rinderpest. We thought that they could do even more and help our fight with the cancer."
Tapping immunological memory and viral platform
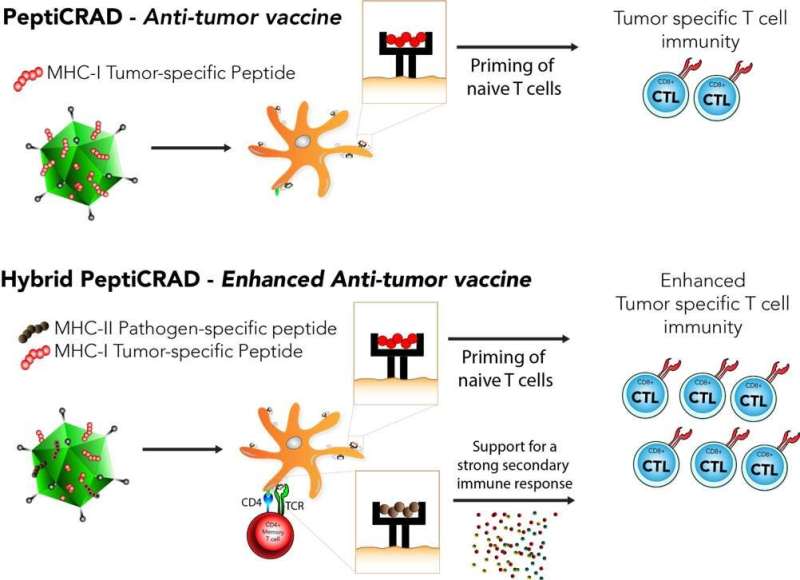
Cerullo and his research team are developing therapeutic cancer vaccines based on viral platform. One of these is called PeptiCRAd, basically a virus dressed as tumors. Cerullo's team generated PeptiCRAd by attaching small tumor pieces (peptides) to the surface of the virus to direct the immune response against the tumor.
"We decided to modify the well-established PeptiCRAd platform in our lab by adding another set of peptides derived from pathogens, which patients have been vaccinated for e.g. tetanus or diphtheria," says Cerullo.
"The main idea behind this new hybrid PeptiCRAd is taking advantage of the pre-existing pathogen-specific immunological memory present in the worldwide population of vaccinated individuals. This is to boost the anti-tumor response directing the pre-existing memory T cells towards the tumor" summarizes Sara Feola, postdoctoral researcher in Cerullo´s lab and one of the co-authors of the study.
More information: Siri Tähtinen et al. Exploiting pre-existing immunity to enhance oncolytic cancer immunotherapy, Cancer Research (2020). DOI: 10.1158/0008-5472.CAN-19-2062