A new strategy to trigger ferroptosis in target cancer cells using drug-metal coordination complexes
Rapidly increasing cancer incidence and mortality worldwide have raised great challenges for the currently available anticancer options, which warrants the development of new therapeutic modalities based on novel antitumor mechanisms. Ferroptosis, a recently discovered form of non-apoptotic cell death, is one such candidate and has already demonstrated immense potential in clinical oncology as it provides alternative therapeutic opportunities for the management of treatment-resistant tumors. In a research study published in Science Advances, lead author Prof. Shu-Hong Yu from the University of Science and Technology of China and his collaborator Prof. Zhong Luo in Chongqing University reported a novel strategy to induce ferroptosis in target cancer cells and further amplify ferroptotic damage for efficient tumor therapy. The nanoformulation was designed to be exclusively activated in the tumor microenvironment, which has demonstrated potent inhibition effects against multiple types of tumors while sparing healthy cells and tissues.
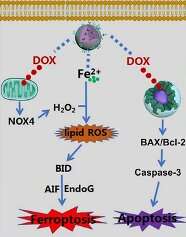
"Overloading tumor cells with ferrous ions could readily initiate the ferroptotic death cascade, and complexed doxorubicin may further amplify the ferroptotic damage by providing additional reactive oxygen species to sustain lipid peroxidation," Says Prof. Shu-Hong Yu, head of the research group at the University of Science and Technology of China. "The benefit of coordinating Fe2+ ions with doxorubicin is manifold. It could not only enhance the stability of Fe2+ ions in biological environment, but also facilitate the subsequent lipid peroxidation process to promote ferroptosis. Moreover, doxorubicin is an FDA-approved anticancer drug capable of inhibiting topoisomerase 2 in tumor cells to prevent DNA replication, leading to a complementary ferroptosis/apoptosis effect against a broad spectrum of tumor indications," Says Prof. Luo, head of the research group at Chongqing University.
The underlying molecular mechanism for the synergy between Fe2+ and doxorubicin is that doxorubicin could generate high level of intracellular ROS by activating the intracellular NADPH oxidase 4 (NOX4) in tumor cells, which may supply H2O2 to sustain the Fe2+-catalyzed lipid peroxidation.
Prof. Shu-Hong Yu also commented on one of the major challenges for the tumor targeted delivery of the Fe2+-doxorubicin complex. "The coordination complex is rapidly dissociated into free Fe2+ and doxorubicin under acidic conditions. However, both species must interact with intracellular components to take effect, necessitating further refinement of the drug delivery process."
Inspired by the recent advances in self-assembly technology, researchers from the two groups developed an intricate self-assembly-based nanoplatform for the tumor-targeted cytosolic delivery of the Fe2+-doxorubicin complex. The Fe2+-doxorubicin complex were efficiently encapsulated into amorphous calcium carbonate nanospheres in a simple one-step co-condensation process, and the surface of the drug-loaded amorphous calcium nanoparticles was modified with polyamidoamine (PAMAM) dendrimer-based tumor-microenvironment-activatable multifunctional ligands, which would remain bioinert during circulation but switch to a tumor-affinitive state upon entering the matrix metalloproteinase-2 (MMP-2)-rich tumor microenvironment.
Thanks to the acid sensitivity of the amorphous calcium carbonate contents, the nanoparticle could be readily degraded in the acidic tumor lysosomes to release the Fe2+-doxorubicin complex, which could be further reverted into free doxorubicin and Fe2+ through the protonation-induced dissociation. Meanwhile, the PAMAM dendrimers could disrupt the lysosomal membrane via a 'proton sponge' effect and release doxorubicin and Fe2+ to the cytosol, where the H2O2 produced during doxorubicin metabolism could stimulate the ferroptotic toxicity of Fe2+ ions to tumor cells.
"Our two groups have been collaborating closely on the synthesis and functionalization of biocompatible inorganic nanomaterials," says Prof. Zhong Luo, head of the research group in Chongqing University. "Amorphous calcium carbonate nanoparticle is a very promising inorganic nanomaterial for biomedical applications. It's easy to synthesize and has tunable drug loading capability. It could also be rapidly degraded in the human body and the degradation products are all non-toxic."
Dr. Menghuan Li, a senior scientist in Prof. Zhong Luo's research group, says that the nanoplatform is also a good example of repurposing old drugs for new applications. "This would greatly benefit the clinical translation of the reported nanoformulation," says Dr. Menghuan Li.
In the future, Prof. Shu-Hong Yu and Prof. Zhong Luo hope to further simplify the synthesis procedures of the amorphous calcium carbonate-based nanoplatform and thoroughly investigate their efficacy and safety in a clinically relevant context. "This system may open up new avenues for treatment against tumors that are resistant to conventional therapies," says Prof. Shu-Hong Yu.
More information: Tumor microenvironment-activatable Fe-doxorubicin preloaded amorphous CaCO3 nanoformulation triggers ferroptosis in target tumor cells Science Advances (2020). advances.sciencemag.org/content/6/18/eaax1346