Cold plasma against the coronavirus
Many seriously ill COVID-19 patients must contend with more than just the coronavirus. Approximately half of the people who were mechanically ventilated yet still died had acquired additional infections in hospital. Cold plasma therapy could prevent these superinfections and reduce the risk of hospital staff becoming infected with coronavirus. Preliminary tests carried out by terraplasma medical GmbH, the subsidiary of the Max Planck spin-off terraplasma, suggest that cold atmospheric plasma (i.e. weakly ionized air) can render SARS-CoV-2 harmless in cell cultures. In order to clarify whether cold plasma can actually help treat COVID-19, investigations in both cell cultures and COVID-19 patients have been initiated together with various partners.
Plasma is the fuel of the stars. In a highly diluted cold variant, ionized gas—or more precisely, ionized air—inactivates bacteria in chronically infected wounds. As studies, conducted with various partners by Julia Zimmermann, CMO, have already shown in the past, atmospheric plasma can also inactivate viruses, like noro- and adenoviruses in solution. It could also help treat COVID-19 patients. "The initial tests suggest that cold atmospheric plasma kills corona viruses in solution," says Jens Kirsch, CEO of terraplasma medical. "This is particularly interesting for us because the viruses are also found in fluids—for example, saliva—on mucous membranes."
The researchers from terraplasma medical carried out the first investigations together with a team led by Albrecht von Brunn, a scientist at the Max von Pettenkofer Institute of the Ludwig-Maximilians-University (LMU) Munich. The preliminary results, which suggest that cold plasma can kill the coronavirus, have prompted terraplasma medical to conduct further studies. Together with the Fraunhofer Institute for Toxicology and Experimental Medicine, terraplasma medical will carry out comprehensive analyses how cold atmospheric plasma affects viruses in cell cultures. Terraplasma medical is also planning studies on patients together with hospitals of the University of Regensburg and the LMU. Because the researchers can build on previous investigations, the clinical trials are possible without the usual animal experiments. "We already know that cold plasmas do not damage the mucous membranes if we use the correct plasma design and the dose does not exceed certain limits," says Gregor Morfill, former Director of the Max Planck Institute for Extraterrestrial Physics, founder of terraplasma, and scientific consultant of terraplasma medical.
Can cold plasma also prevent the virus from spreading to the lungs?
By conducting the clinical trials, the researchers hope to answer various questions. They want to clarify whether treatment with cold plasma can prevent mechanically ventilated COVID-19 patients from becoming infected with hospital germs (thus considerably improving the chances of a cure) as well as whether ionized air significantly reduces the viral load in the mouth, nose, and throat of these patients. This could additionally help to reduce the risk of infection for medical staff in intensive care units. "We hope that in the long term we will also be able to prevent the virus from spreading from the mouth, nose, and throat to the lower respiratory tract of COVID-19 patients whose lungs are still free of the virus," says Kirsch. "We could thus reduce the number of COVID-19 patients requiring treatment in intensive care units or mechanical ventilation."
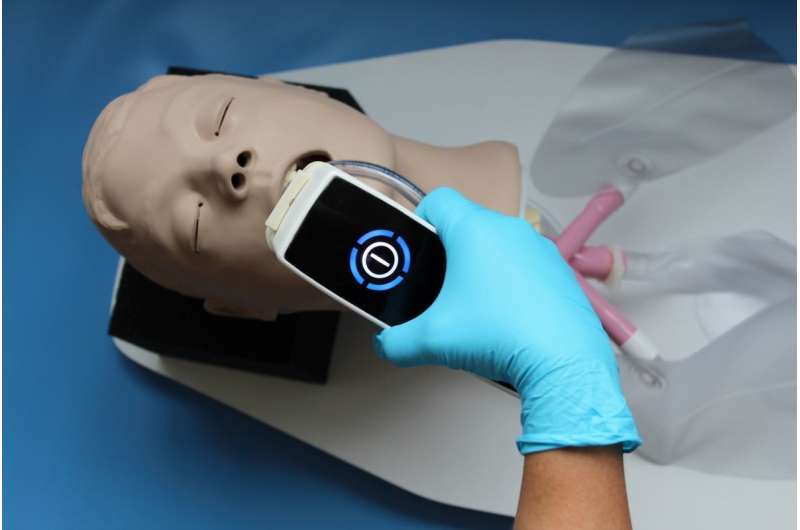
In order to be able to use the cold plasma in the upper respiratory tract, terraplasma medical has converted a device used to treat chronically infected wounds. However, before doctors can treat the first COVID-19 patients, the clinical trials must first be approved by the respective ethics committee. "We expect the first results in six to seven months," says Gregor Morfill.