Repurposed drug tested in COVID-19 trial, thanks to Cancer Research UK know-how
Cancer Research UK and its partners have today launched a clinical trial to test if a drug that's used to treat inflammation of the pancreas can help people with COVID-19.
Cancer patients have been severely affected by COVID-19, impacting on how they are diagnosed, treated and cared for. Beating cancer remains Cancer Research UK's priority, but the charity realises that it will not be able to fully focus on its mission until COVID-19 itself is under control and where possible, has been supporting the national effort, while still pushing forward with its mission to beat cancer.
The involvement of Cancer Research UK's Centre for Drug Development (CDD) in the new SPIKE1 trial is an example of where the charity has been able to work with industry and academia. Rapidly responding and contributing to the COVID-19 effort, without redirecting efforts from cancer patients with the CDD still able to continue treatment for all cancer patients on their phase I trials.
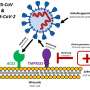
The SPIKE1 trial, funded by LifeArc, is a partnership between the CDD, Latus Therapeutics and the University of Edinburgh. It will investigate whether a drug called camostat, which has been shown in the lab to prevent COVID-19 from entering human cells, could help control symptoms of the virus and prevent people that are infected needing to go to hospital. Camostat is already licensed in Japan and South Korea to treat pancreatic disease, so if successful it could be quickly manufactured and used to treat people with COVID-19.
Almost half of UK COVID-19 patients requiring critical care have died in hospital so far. Currently, there is a limited number of treatment options available for people affected by COVID-19, such as the recently approved antiviral drug remdesivir.
Scientists around the world are racing to find a vaccine against the killer virus, but even with the impressive progress seen so far, it will take time before an effective vaccine can safely be given to people. While the world waits for a vaccine, repurposing already available drugs such as remdesivir or camostat to treat COVID-19 could more quickly save lives.
Realising this, Dr. Bobojon Nazarov, founder of biotech company Latus Therapeutics, while in lockdown, identified key studies showing that camostat prevents coronavirus from entering the host cell. He was keen to quickly set up a clinical trial to see if camostat could be used to effectively treat people with COVID-19.
Working with a team of scientists and clinicians from the Universities of Oxford and Edinburgh, he approached the team at the CDD who, realising the potential of camostat, adapted their processes, expedited protocol development and set up activities to help get the trial off the ground.
The CDD team together with Latus Therapeutics also navigated the complex logistics while the world was moving towards lockdown and quickly secured shipping of camostat from Ono Pharmaceutical Co., Ltd., the company that makes it in Japan.
Latus Therapeutics was instrumental in setting up the research project and securing £1m funding from LifeArc for a clinical trial to test if camostat is effective at treating patients with COVID-19. The project will be sponsored and managed by the CDD at no cost to Cancer Research UK.
Iain Foulkes, executive director of research and innovation at Cancer Research UK, said: "We're seeing the impact of COVID-19 on cancer patients throughout the country and we have the skills at Cancer Research UK to assist the national effort in helping to beat this virus, and support from LifeArc is critical to this new trial.
"The charity's Centre for Drug Development has a strong track record in setting up trials quickly, which is a testament to our sector leading ways of working. The team have shown that in these uncertain times they've not only managed to continue treatment for all cancer patients on their phase I trials, but also excelled at this challenge and lent their expertise to others in need. Because we know that the sooner we can find ways to minimise the impact of COVID-19, the more quickly we can more fully return to our life saving cancer research."
Melanie Lee, LifeArc's CEO, said: "As an independent medical charity with expertise in medical translation, LifeArc could rapidly offer the resources to evaluate proposals and financially support studies with the best chance of improving patient outcomes. We are pleased to support this vital work during the COVID-19 pandemic."
Camostat has a proven safety record because it's already used to treat patients with chronic pancreatitis, a disease that causes severe pain because of an inflamed pancreas. This meant it could proceed straight to a phase III clinical trial, to see if it is effective against COVID-19.
The trial will take place in the community, recruiting people with symptoms of COVID-19 that don't yet need hospital care. Those receiving treatment will take daily doses of the tablet and all patients will be assessed daily by telephone and self-report their temperature and blood oxygen levels. The objective of the trial is to see if taking camostat early after COVID-19 symptoms begin can stop the infection from getting worse, reducing the need to go to hospital.
Dr. Bobojon Nazarov, founder of Latus Therapeutics, said: "Camostat belongs to the only class of drug that has a strong mechanistic basis for blocking entry of the virus into human cells. We believe this drug could be used to reduce the severity of COVID-19 infection, providing much needed time for the body's immune system to recognise the virus and destroy it. Unlike finding a vaccine, this drug could be used quickly to help people recover from COVID-19."