Antibiotics in early life slows digestive nerve function, alters microbiome
A new study finds antibiotic exposure during crucial developmental periods in early childhood alters digestive tract nerve function and bacterial colonies. The study is published in the American Journal of Physiology-Gastrointestinal and Liver Physiology. It was chosen as an APSselect article for July.
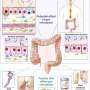
The gastrointestinal tract contains the largest community of bacteria and other microorganisms in the body (gut microbiome), as well as an extensive network called the enteric nervous system. Together, the gut microbiome and the enteric nervous system control digestion, gut movement, fluid exchange and blood flow to the digestive system. The enteric nervous system and gut microbiome continue to develop after birth and into the first months of life, in part due to the transition to solid food that occurs during this time.
Research has shown that antibiotics affect gut microbiota by changing the populations of different bacteria. Antibiotic use in early life, particularly in premature infants, has been tied to increased risk of developing metabolic disease, obesity and allergies. In a new study, researchers explored the effects of antibiotic exposure on the gut microbiome and the enteric nervous system as it related to age.
The research team studied two groups of mice—one was newborn (neonatal) and the other was recently weaned—that were exposed to the antibiotic vancomycin. Vancomycin is used to treat a wide variety of bacterial infections, including inflammation of the intestines (colitis). The treated mice were compared with age-matched controls.
Vancomycin treatment induced dysbiosis in both treated groups of mice. Dysbiosis is defined as an alteration in the gut microbiome that can lead to disease. But the antibiotic had different effects on microbiota composition of the two age groups. This is in part due to the significant maturation of microbiota between the neonatal and weaning periods.
The weaned mice exposed to antibiotics had changes to the nerve cells in the enteric nervous system and slower colonic migrating motor complex patterns. These patterns of electrical activity prompt the muscles of the colon to propel stomach contents through the digestive tract. Intestinal transit time is linked to gut microbiota composition and diversity, and slow transit time is often a cause of constipation.
The study's results support the idea that "antibiotics administered during critical developmental windows have greater consequences on host physiology than antibiotic exposure during maturity," the researchers wrote. Changes in communication between the microbiota and the enteric nervous system that arise from early-life antibiotic treatment could have lasting effects on gastrointestinal function.
More information: Lin Y. Hung et al. Antibiotic exposure postweaning disrupts the neurochemistry and function of enteric neurons mediating colonic motor activity, American Journal of Physiology-Gastrointestinal and Liver Physiology (2020). DOI: 10.1152/ajpgi.00088.2020