After chronic stress in adolescence, male and female reactions to trauma differ later in life

Eighteen months ago, eight female researchers in the lab of Gretchen Neigh, Ph.D., at Virginia Commonwealth University began studying how early life experiences and exposure to chronic stress can have lingering effects into adulthood. They discovered that stressors occurring early in life, such as violence or child abuse, may "shape your brain in ways you don't expect," said lead author Gladys Shaw, a Ph.D. candidate in the Biomedical Sciences Doctoral Portal.
The research team found that two individuals exposed to the same stress as adults can react differently if one had experienced early life trauma. Stressors or trauma in adolescence can shape "the way your biology will respond to a car accident when you're older, for instance," said Neigh, an associate professor in the Department of Anatomy and Neurobiology at the VCU School of Medicine.
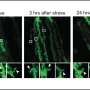
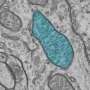
The research focused on the role inflammation plays in the stress response as well as the stress- and inflammation-induced changes that may affect mitochondrial function in the brain. Mitochondria produce chemical energy that powers biochemical reactions in cells.
Their findings were published May 7 in Brain, Behavior, and Immunity, the journal of the PsychoNeuroImmunology Research Society.
Looking at chronic stress and inflammation
"In this research, we are trying to understand what exactly is changing the signals that are carried forward," Neigh said. "We are trying to figure out how we can intervene and how can we fix potentially harmful or negative signals even if a critical window has passed."
The team's preclinical research encompassed exposure to chronic stress during both adolescence and early adulthood, chronic adult inflammation, and a combination of the two.
When exposed to chronic stress or chronic inflammation, similar to a trauma situation like a car wreck, male and female subjects reacted differently, but when exposed to both together, all subjects experienced lasting effects on the brain.
"In the males, individual exposures to trauma or inflammation did not alter mitochondria in brain cell connections, but the combined exposure compromised the ability of the mitochondria in brain connections to provide the normal amount of fuel to the cells," Neigh said.
The team discovered that the same stressors can affect both sexes, but the impact is different according to biological sex, scientifically defined as the hormonal and chromosomal profile of an organism.
"This 'two-hit' hypothesis is extremely interesting when we think about disease progression across the lifespan. When an inflammatory disease, something like COVID-19, hits someone after a life of chronic stress, that second event tips the scales," said Molly Hyer, Ph.D., a postdoctoral fellow in Neigh's lab.
The paper being published is "exciting for a number of reasons," Hyer said.
"The whole concept is a novel idea of looking at how stress and inflammation shift brain metabolism by altering oxygen consumption and mitochondrial function," she said. "The effect of stress we observed here is unique and sets the stage for understanding how adolescent stress can drive lifelong changes in neural function."
"It's important to look at the differences in sexes when you are looking at disease," Hyer said. "While males and females both have a stress response, an immune response, hormones, etc., science is clearly showing that the way these systems interact is unique to each sex. This means that diseases manifest differently in men and women. The best example, and very relevant for this project, is that women are twice as likely as men to develop stress-induced disorders like depression and [post-traumatic stress disorder]."
Representing the underrepresented
For many years, treatments did not reflect the female physiology. As recently as this spring, doctors have spoken out about the importance of considering sex differences in medicine from a perspective broader than just reproductive health.
"Our medical model has been based on knowledge of male subjects for centuries, and making our educational and research models more inclusive through the work of teams like Dr. Neigh's is crucial to improving the understanding of how our brains work and how our bodies function," said Peter Buckley, M.D., interim CEO of VCU Health System, interim senior vice president of VCU Health Sciences and dean of the School of Medicine.
In 2015, the National Institutes of Health put out a mandate requiring the inclusion of females in scientific research to combat the lack of data.
"Dr. Neigh and I have both worked with males and females throughout our research careers and have seen how important it is to consider sex as a biological variable when you are studying ways to treat psychiatric disease," Hyer said.
"With females, if we find out that hormones influence stress-specific changes in mitochondrial function, then we can figure out who needs what kind of treatment and tailor pharmaceuticals to the individual," she said, adding she also wants to look into the aging aspects as sex hormones decrease. "This may help us treat and prevent anxiety and depressive disorders as well as age-related disorders such as Alzheimer's and Parkinson's."
Shaw, the paper's lead author, hopes the research will help determine who is more susceptible to a specific environmental stressor.
"These data highlight that different mechanisms are likely in play between the sexes and that sex differences in neural outcomes may be precipitated by sex-specific effects of life experiences" on the way cells behave, Shaw concludes in the paper.
The data from this study indicates that males and females use energy in the brain differently after they have experienced stress or inflammation.
"Energy usage in the brain can have a cascade of impacts on neural function—both cognitive and behavioral," Hyer said. "That so many psychiatric diseases may be related to energy usage in the brain, it is essential that we understand this puzzle piece within the greater picture."
More information: Gladys A. Shaw et al. Traumatic stress history interacts with sex and chronic peripheral inflammation to alter mitochondrial function of synaptosomes, Brain, Behavior, and Immunity (2020). DOI: 10.1016/j.bbi.2020.05.021