Researchers find three distinct immune responses for sicker COVID-19 patients

Researchers from the Penn Institute of Immunology discovered three distinct immune responses to the SARS-CoV2 infection that could help predict the trajectory of disease in severe COVID-19 patients and may ultimately inform how to best treat them.
The findings were published in Science.
"For patients who are hospitalized with COVID-19, there isn't just one way for the immune system to respond. There's a lot of heterogeneity, which we've distilled down into what we're calling three 'immunotypes,' said senior author E. John Wherry, Ph.D., chair of the department of Systems Pharmacology and Translational Therapeutics and director of the Penn Institute of Immunology in the Perelman School of Medicine at the University of Pennsylvania. "We're hopeful we may actually be able to predict, or at least infer, the different immune patterns a patient has based on clinical data. This would allow us to start thinking about enrolling patients to different types of clinical trials investigating treatments."
The coronavirus triggers different immune responses and symptoms in critically ill patients, but how those two correspond has remained poorly understood, making treatment decisions more difficult.
While recent studies reveal details on the immune's response to the virus, most have been single-case reports or focused on a small group of individuals. This is the first study, to the author's knowledge, to offer up a comprehensive immune profile of a large number of hospitalized patients.
The researchers applied deep immune profiling to capture individual responses of 163 patients during the course of their infections. The study included 90 hospitalized patients treated at the Hospital of the University of Pennsylvania, 29 non-hospitalized patients, and 44 healthy donors with no COVID-19 infection. The immune responses varied among the group, but there were patterns that hold clinical promise.
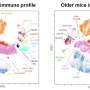
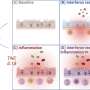
The first immunotype had robust CD4+ T cell activity, with modest activation of CD8+ T cells and peripheral blood lymphocytes. CD4+ and CD8+ act as the main inflammatory immune cells that work to clear viruses. The second immunotype was characterized mainly by a subset of CD8+ T cells known as EM and EMRA and a modest activation of CD8+ T cells, memory B cells, and peripheral blood lymphocytes. The third immunotype showed little to no evidence of an immune response to the infection.
Next, researchers combined the profiling with clinical data to understand the relationships between immune responses and disease. The first immunotype was tied to more severe disease that included inflammation, organ failure, and acute kidney disease. The second correlated not with disease severity but instead pre-existing immunosuppression and mortality. The third type, which had no immune activation, was not associated with specific symptoms or clinical features, though they varied.
The immunotypes developed by Wherry and team represent adaptive immune responses. A second study from researchers at Penn, published in Science Immunology, uncovered new details about the innate, or initial, response to SARS-CoV2.
"T and B cell activity are informed by innate immune responses," said senior author Michael R. Betts, Ph.D., a professor of Microbiology and program leader in the Penn Institute of Immunology, who is also a co-author on the first study. "We believe what's happening with the innate response of the immune system might be what's leading to these three immune phenotypes Dr. Wherry's lab identified."
Profiling the blood samples of 42 infected patients (with moderate and severe disease) and 12 healthy donors, the researchers found a similar heterogeneity in immune adaptive responses: robust activation of CD4+ and CD8+ T cells, B cells, along with peripheral blood cells, like neutrophils, monocytes, and "natural killer," or NK, cells.
While the innate responses were also heterogenous, the researchers observed a decrease of CD15 and CD16 molecules on neutrophils and CD16 on NK cells, immature granulocytes, and monocytes, in patients with more severe disease. These two molecules are known players in the immune's response to viral infections that also represent a potential target for immunotherapy. How they are driving and exacerbating the adaptive responses in the three immunotypes is an important question the labs are working to better understand.
COVID-19 studies have been moving at an unprecedented speed as researchers band together to find answers. Among its many efforts, Penn formed lab and clinical research teams from diverse backgrounds to strengthen its focus on the immune system, along with the COVID Processing Unit to manage specimens to profile.
"Understanding the power of the immune system to regulate responses to disease is one of the major advances in medicine in the last decade, and Penn has been at the center leading that discovery. We are now applying the broad expertise and experience of our more than 200-person immunology community toward the research and treatment of COVID-19," said Jonathan A. Epstein, MD, executive vice dean, chief scientific officer, and a professor of Cardiovascular Research at Penn. "The deep immuno-profiling work the investigators applied here is likely to be useful not only now, for this disease, but into the future for many others."
More information: Mathew Divij et al, Deep immune profiling of COVID-19 patients reveals distinct immunotypes with therapeutic implications,Science 15 Jul 2020 DOI: 10.1126/science.abc8511