New reporter mouse strain offers powerful genetic tool to identify P2X2-expressing cells
Despite frequent news announcing "medical breakthroughs," advancements in biomedical and clinical science typically happen incrementally. Scientists refine our understanding of how the world works by harnessing new tools and data that can challenge conventional thinking—a continual process of revision that elicits new answers to old questions, and often poses different questions.
In an eNeuro paper published July 15, University of South Florida Health Morsani College of Medicine researchers describe a reporter mouse strain they created in pursuit of a new way to answer an old question: Is purinergic receptor gene P2X2 expressed in particular populations of sensory nerve cells?
"We needed a suitable mouse model to visualize where P2X2 is located so we might prove the gene is actually expressed in a very discrete group of sensory nerves. And because, moving forward, we want a reporter system that allows us to manipulate these vagal nodose nerves in precise, varied ways for therapeutic purposes," said senior author Thomas Taylor-Clark, Ph.D., a professor in the Department of Molecular Pharmacology and Physiology.
"This paper is an example of how reexamining questions with better techniques leads to clearer understanding, and in this day and age the clarity and reproducibility of data is a paramount issue in science."
The P2X2 receptor (P2X2 for short) belongs to a family of P2X ion channels that sit on the surface of cell membranes and are activated by the neurotransmitter adenosine triphosphate (ATP). P2X2 plays a key role in sensory processes, including taste, hearing, some aspects of blood pressure regulation, and sensing physical stimuli in visceral organs like the lungs and bladder.
Dr. Taylor-Clark studies airway sensory nerves affecting defensive behaviors, including cough, and what happens when they go wrong in disease and injury. To further their research, his team needed a more reliable approach to distinguish which subsets of cells express P2X2, especially in the brain and spinal cord (central nervous system) and the peripheral nervous system (nerves outside the brain and spinal cord). Existing pharmacological and biochemical techniques were not selective enough, yielding dramatically different gene expression patterns that hamper accurate estimates of P2X2-expressing cell types.
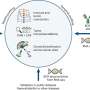
So, the USF Health researchers created a knockin mouse incorporating a powerful genetic approach that could be used in future experiments. They made a mouse that expresses the bacterial enzyme cre recombinase in cells expressing the P2X2 gene. The enzyme manipulates specific sites (lox sequences) in DNA. Then, they bred this P2X2-cre mouse with a second mouse having specific lox sequences that produce substantial levels of tdTomato—a bright red fluorescent protein—under the control of cre. In offspring of the P2X2-cre mice and the cre-sensitive mice, tdTomato is robustly expressed and specifically reported (visualized) in P2X2-expressing cells, even when levels of P2X2 expression are low.
"With this system, it's easier to see any cell type you want to investigate," Dr. Taylor-Clark said. "And, since many mouse strains have different cre-sensitive genetic expression patterns, you can manipulate virtually any gene or genetic process to test its role in tissue/organ function with a modular approach."
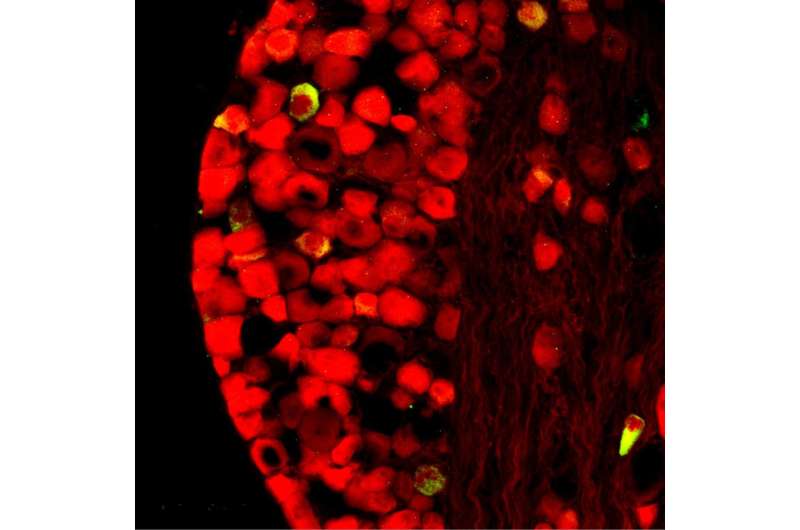
The researchers detailed where they found P2X2. As they suspected, the gene was expressed predominantly in the vagal sensory nerve system, where cell clusters relay sensory information about the state of the body's organs to the central nervous system. In particular, almost all nodose vagal neurons (more than 85%) expressed P2X2, compared to nearly none of the jugular neurons. (Nodose and jugular are the two groups of neurons in the vagal system.).
The researchers demonstrated some P2X2 expression in the tongue's taste buds, the carotid body, trachea (windpipe) and esophagus. They observed P2X2 in hair and support cells of the cochlea (inner ear bone important in hearing), but not, as some previous studies reported, in sensory nerves innervating the hair cells.
With a few exceptions, P2X2 expression was absent in central nervous system cell types. Earlier reporter mouse studies using established biochemical techniques indicated P2X2 expression in virtually every area of the brain, so the USF Health group was surprised to find P2X2 expressed in a very limited subset of neurons, Dr. Taylor-Clark said.
"But, actually, that was encouraging because if we manipulate (gene expression) we want the effects to be very narrow and targeted, not widespread," he added. "Selectivity is the hallmark of any therapeutic approach. Otherwise, you will not get the beneficial outcome you want, and you may get side effects you don't want."
Other studies have suggested that activating nodose sensory nerves diminishes cough, while activating jugular sensory nerves increases cough. Dr. Taylor-Clark hopes to test whether nodose neurons can protect against chronic cough by modifying the P2X2-cre system to selectively silence only the nodose neurons, without adversely blocking all other nerve impulses.
"Our next step is to manipulate this P2X2-cre system so that, instead of expressing tdTomato, we can express a protein that upon addition of a drug then either artificially activates or inhibits P2X2-expressing cells," he said. "Currently, little is understood about the physical interaction of the nodose nerve terminals (endings) in the trachea and other target organs, and how that changes with disease. Our goal is a detailed knowledge of all the different subtypes of sensory nerves and how they control organ function, so we can help drive targeted neuromodulaton therapies."
More information: Seol-Hee Kim et al, Development of a mouse reporter strain for the purinergic P2X2 receptor, eNeuro (2020). DOI: 10.1523/ENEURO.0203-20.2020